
Get answers about treatment, top doctors, and clinical trials from the nation’s most trusted mesothelioma resource.
Get Your Free GuideCAR T-cell therapy is an experimental treatment for mesothelioma. It's a type of immunotherapy. It modifies a person's T cells to target a protein on mesothelioma cells called mesothelin. It's offering hope for future treatment of this aggressive cancer.
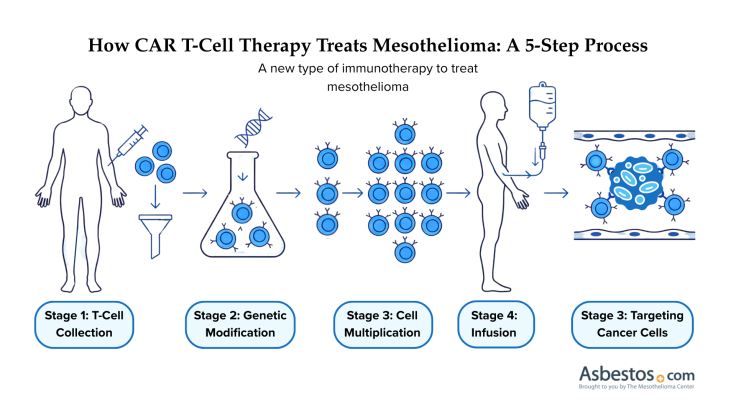
A new experimental way to treat mesothelioma is a type of immunotherapy called CAR T-cell therapy. This treatment uses a person’s own immune cells, known as T cells, to fight the cancer. Doctors take some of the person’s T cells and change them in a lab.
They give the cells new instructions so they can make something called a chimeric antigen receptor. This special receptor, or CAR, helps the T cells find and attack cancer cells. CAR T-cell therapy is a type of adoptive cell transfer. This means it uses a person’s own cells to help fight cancer.
CAR T-cell therapy is already approved for some blood cancers like leukemia and lymphoma. But for mesothelioma, it’s still being tested and is only available in clinical trials. Researchers hope that helping T cells better find mesothelioma cells will improve outcomes for people in the future.
CAR T-cell therapy helps T cells target mesothelin, a protein mainly found in mesothelioma. This allows them to distinguish cancer cells from healthy ones.
Boosting Immunity Against Mesothelioma
After adding the CAR, doctors grow T cells in a lab until there are millions of them. Then, they’re injected back into your body through a blood infusion. After reinfusion, they circulate and attach to mesothelioma cells via their new receptor, like a magnet finding metal.
This binding acts like a key turning a lock. It activates the T cells to release toxic chemicals that destroy the cancer cell. Additionally, CAR T cells can signal and recruit other parts of the immune system to help in the fight.

Get answers about treatment, top doctors, and clinical trials from the nation’s most trusted mesothelioma resource.
Get Your Free GuideCAR T-cell therapy for mesothelioma is still experimental and only accessible through clinical trials. It’s not FDA-approved for this use. No cancer centers provide it outside of research settings. You must join a clinical trial to receive this therapy.
While the therapy is complex and requires several weeks from start to finish, each step is carefully coordinated to maximize your safety and treatment success. Understanding the CAR T-cell therapy process can help you and your family prepare.
The CAR T-Cell Therapy Process
You’ll stay under close medical care for several weeks after the infusion. Side effects can happen right after treatment or sometime later. Careful observation and rapid treatment are critical. CAR T-cell therapy for mesothelioma is still experimental, but it allows trial participants to contribute to research and benefit from new immunotherapy advancements.
Like any potent mesothelioma treatment, CAR T-cell therapy can cause side effects. These can include fever, chills or fatigue. Because it supercharges the immune system, many of the side effects are related to an overactive immune response.
Cytokine release syndrome is a common side effect of CAR T therapy. When CAR T cells activate, they release many chemical signals called cytokines. Too many cytokines can lead to a strong immune reaction. If severe, CRS can lead to dangerously low blood pressure, rapid heartbeat, trouble breathing and organ dysfunction.
Common CAR T-Cell Therapy Side Effects
People receiving CAR T-cell therapy in trials are closely monitored for adverse effects, particularly in the initial days and weeks post-infusion. Some people experience mild symptoms, while others face serious complications.
CAR T-cell therapy can affect the nervous system. People may experience neurological symptoms like sleepiness, confusion or agitation. Doctors and nurses will manage any issues. Most side effects can be treated effectively if addressed promptly.

Connect with top-rated mesothelioma specialists at a cancer center near you, who will personalize treatment options based on your diagnosis.
Find Your CenterClinical research is shaping the future of CAR T-cell therapy for mesothelioma. Ongoing trials aim to enhance CAR T-cell design, delivery and effectiveness against tumors. Although results have been promising, many people with mesothelioma still encounter challenges. These include immune resistance and tumor walls that are hard for treatments to cross.
One major clinical trial (NCI-2024-01620) is testing an advanced, experimental type of cell therapy (an autologous logic-gated Tmod™ CAR T-cell product) in subjects with solid tumors, including mesothelioma. This study, happening at the National Cancer Institute, offers patients a new way to fight their cancer using immunotherapy.
Goals of Active CAR T-Cell Therapy Clinical Trials
Researchers at Houston Methodist and Weill Cornell Medicine developed CAR T cells that better target mesothelin. A human trial evaluated a new CAR T-cell design with different message pathways (like DAP12) to target mesothelin. It showed early signs of working safely, like shrinking tumors or stopping growth.
Studies show there are many ways to improve CAR T-cell treatments for mesothelioma. Early results indicate using local delivery, targeting 2 proteins and boosting the immune system can help solve current problems. With continued progress, CAR T-cell therapy may become a long-term treatment option for people with mesothelioma.
Recommended ReadingYour web browser is no longer supported by Microsoft. Update your browser for more security, speed and compatibility.
If you are looking for mesothelioma support, please contact our Patient Advocates at (855) 404-4592
The Mesothelioma Center at Asbestos.com has provided patients and their loved ones the most updated and reliable information on mesothelioma and asbestos exposure since 2006.
Our team of Patient Advocates includes a medical doctor, a registered nurse, health services administrators, veterans, VA-accredited Claims Agents, an oncology patient navigator and hospice care expert. Their combined expertise means we help any mesothelioma patient or loved one through every step of their cancer journey.
More than 30 contributors, including mesothelioma doctors, survivors, health care professionals and other experts, have peer-reviewed our website and written unique research-driven articles to ensure you get the highest-quality medical and health information.
My family has only the highest compliment for the assistance and support that we received from The Mesothelioma Center. This is a staff of compassionate and knowledgeable individuals who respect what your family is experiencing and who go the extra mile to make an unfortunate diagnosis less stressful. Information and assistance were provided by The Mesothelioma Center at no cost to our family.LashawnMesothelioma patient’s daughter


Asbestos.com. (2026, March 9). CAR T-Cell Therapy for Mesothelioma. Retrieved March 27, 2026, from https://www.asbestos.com/treatment/immunotherapy/car-t-cell-therapy/
"CAR T-Cell Therapy for Mesothelioma." Asbestos.com, 9 Mar 2026, https://www.asbestos.com/treatment/immunotherapy/car-t-cell-therapy/.
Asbestos.com. "CAR T-Cell Therapy for Mesothelioma." Last modified March 9, 2026. https://www.asbestos.com/treatment/immunotherapy/car-t-cell-therapy/.

Dr. Landau is the Medical Director of Virtual Hematology at the Medical University of South Carolina, where he leads programs that expand access to cancer care through telehealth. With more than 18 years of experience in oncology and hematology, he specializes in hematologic and genitourinary cancers, including bladder, prostate and kidney cancers. He has held multiple leadership roles in cancer program development and previously served as section chief of hematology and oncology at Orlando Health UF Health Cancer Center, where he founded its telehealth program.
Our fact-checking process begins with a thorough review of all sources to ensure they are high quality. Then we cross-check the facts with original medical or scientific reports published by those sources, or we validate the facts with reputable news organizations, medical and scientific experts and other health experts. Each page includes all sources for full transparency.
Please read our editorial guidelines to learn more about our content creation and review process.
