
Access mesothelioma clinical trials matched to your diagnosis. Get help with eligibility and enrollment.
Get StartedThe p53 tumor-suppressor gene helps the body fight cancer cells. Mutations in this gene are associated with all types of cancer. Gene therapy to fix or replace defective p53 genes is a new way to treat cancers such as mesothelioma.
The p53 gene therapy is an innovative cancer treatment strategy. It involves repairing or replacing a gene that produces the tumor-suppressor protein p53. The p53 protein is often called the “guardian of the genome.” It repairs DNA and prevents uncontrolled cell growth and cancer.
Restoring the body’s ability to produce functioning p53 protein is the goal of p53 gene therapy. Mutations in this gene appear in more than 50% of all cancers. But they are rarely seen in healthy tissue. Targeting p53 may help treat and possibly cure mesothelioma and other cancers.
Therapies to Treat p53 Mutations
Many experts believe p53 gene therapy can treat various cancers, including mesothelioma. Several interventions target p53 mutations. They include gene therapy and drugs that restore or inactivate p53. The drugs aim to fix the mutated p53 genes and proteins.
Different mutations are susceptible to different treatments. Some treatments only work when specific mutations are present. Sometimes, p53 gene mutations cause defective p53 protein to build up in cells. This can lead to cancer. Some treatments aim to degrade these defective proteins.
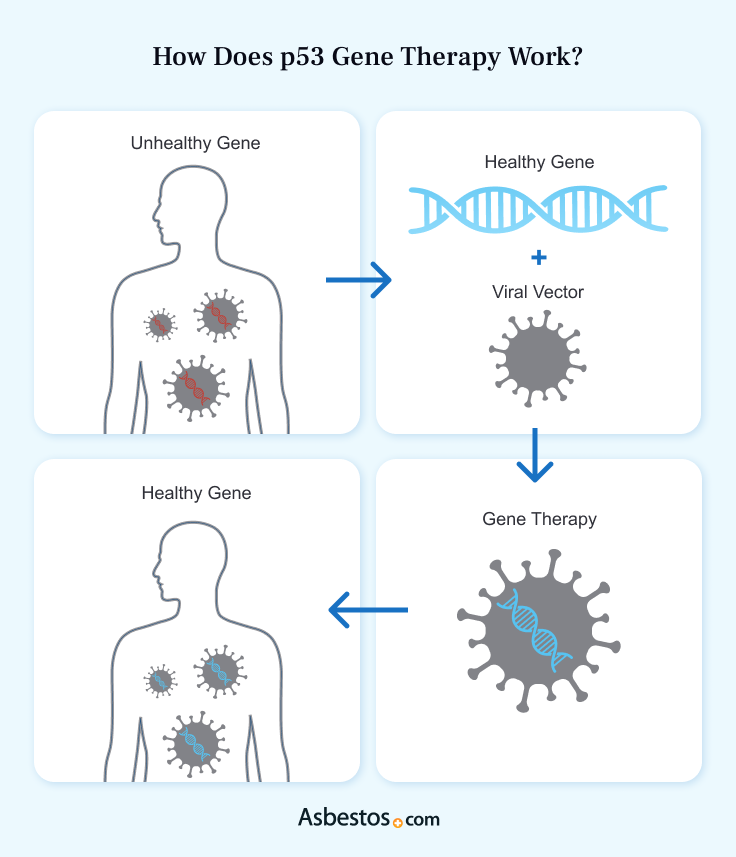
Gene therapy inserts a functioning gene into cells with a mutated gene. The cell’s DNA takes in this gene, allowing it to produce a working protein. The U.S. Food and Drug Administration hasn’t approved many gene therapies. This treatment may be available in clinical trials.
Doctors can safely combine p53 gene therapy with other cancer treatments. For example, doctors may use radiation, chemotherapy, immunotherapy and surgery combos. This may increase your treatment’s effectiveness.
Another form of gene therapy in development for mesothelioma includes suicide gene therapy. Doctors are trying to figure out how to fix or replace damaged p53 genes in cancer cells. It could help treat different types of cancer.
The biggest challenge of gene therapy is getting working genes into cells with faulty ones. Successful treatment requires delivering genes to the right cells. Then, the cells’ DNA must incorporate the genes. There are several methods to achieve this, including using viruses and nanoparticles.
Ongoing trials are testing p53 gene therapy for mesothelioma treatment. For example, researchers are testing it in combination with other treatments.
Genetically engineered viruses can deliver gene therapy to cells. Researchers use several types of viruses as vectors to transfer DNA into human cells. Scientists change these viruses to prevent them from replicating or causing infections.
Doctors can inject these altered viruses into the body, sometimes directly into tumors. This method of delivering gene therapy has proven safe and effective. But it isn’t efficient. These viruses may not reach all cancer cells. The immune system may destroy them before they reach their target.
Oncolytic viruses are genetically altered viruses. They attack cancer cells and spare healthy ones. These viruses can also serve as vectors to carry DNA to cancer cells. Research shows oncolytic viruses can transfer p53 genes to cancer cells. This increases their ability to destroy the cancer cells.
Researchers have used a modified variation of the measles virus to fight mesothelioma. Laboratory studies show oncolytic viral therapy may treat mesothelioma effectively. This is true with or without gene therapy.
Nanoparticles can transfer DNA or other molecules into cells. They’re no larger than 100 nanometers. That’s 1,000 times smaller than a human hair’s thickness.
These nanoparticles are more efficient than viruses for delivering gene therapy to cancer cells. Scientists can design nanoparticles to target only cancer cells. Research suggests nanoparticles may safely restore p53 function in cancer cells.
Some new and existing drugs affect the p53 gene or protein. There are many possible mutations in the p53 gene. Some prevent cells from making p53 protein. Others cause abnormal p53.
Drugs that let cells make a working p53 protein can treat some mutations. These mutations block cells from making this protein. Other drugs can stabilize the p53 protein’s 3-D shape. This shape is vital for the protein to work properly. Several drugs are in trials to test their ability to treat cancer.

Access mesothelioma clinical trials matched to your diagnosis. Get help with eligibility and enrollment.
Get StartedA major benefit of p53 gene therapy is that it can target cancer cells. Other treatments that target p53 can do the same. Cancer drugs can harm healthy cells as well as cancer cells. This causes many side effects. Targeting only cancer cells or cells with p53 mutations may reduce side effects.
Studies show p53 gene therapy can reverse cancer cells’ resistance to radiation. It can also lessen radiation’s side effects. p53 gene therapy works best with other cancer treatments. Examples include radiation, immunotherapy or chemotherapy.
Adding p53 to some tumor cells or activating WT p53 in tumor cells can result in cell death, tumor remission or even cures.
Research shows this therapy is safe and effective for certain cancers with p53 mutations. For example, researchers have tested Gendicine in trials on several cancers. Gendicine is a viral vector containing the p53 gene.
Tests of other p53-based treatments in cells and animals were successful. Researchers will need to conduct human trials as well.
The side effects of p53 gene therapy are typically mild compared to those of chemotherapy or radiation. The most common side effect is flu-like symptoms. They include fever and pain after taking the drug. Gastrointestinal symptoms are also common.
Most reactions to p53 gene therapy are mild. However, serious side effects can occur. These include kidney and liver dysfunction and bone marrow suppression. A rare and serious side effect is platelet crisis. This can cause spontaneous bleeding. It’s a life-threatening condition.
Possible Adverse Reactions to P53 Gene Therapy
Some side effects seen in clinical trials depend on the location of the cancer. For example, treatment can harm the lungs of lung cancer patients. It can also cause urinary problems in cervical cancer patients.

Connect with top-rated mesothelioma specialists at a cancer center near you, who will personalize treatment options based on your diagnosis.
Find Your CenterClinical trials are vital. They test new treatments to ensure they’re safe and effective. There is no FDA-approved gene therapy for mesothelioma. The only way to access this treatment is in a clinical trial. Clinical trials for p53 gene therapy and p53-based treatments are ongoing around the country.
Recruiting Clinical Trials
There are clinical trials for drugs that interact with p53 and MDM2, which regulates p53. Your mesothelioma doctor and Asbestos.com Patient Advocates can help. They can provide info on clinical trials you may qualify for.
Research shows p53 gene therapy may be able to treat certain mesothelioma cases. However, only people with p53 mutations can benefit from p53 gene therapy.
A 2022 research study found p53 mutations in 18% of pleural mesothelioma and 15% of peritoneal mesothelioma cases. This means more than 80% of mesothelioma patients have a functioning wild-type p53 gene. So they may not benefit from this treatment.
Lab research has paved the way for clinical trials. They’ll test p53-based treatments for mesothelioma. Cell cultures show p53 gene therapy can boost chemo for pleural mesothelioma.
Soon, new mesothelioma treatments that target p53 may be in clinical trials. Treatments focusing on p53 and its role in tumor growth may change how we treat mesothelioma and other cancers.
Recommended ReadingStay up-to-date on treatment, research, clinical trials, doctors and survivors
The information on this website is proprietary and protected. It is not a substitute for professional medical advice, diagnosis or treatment. Any unauthorized or illegal use, copying or dissemination will be prosecuted. Please read our privacy policy and terms of service for more information about our website.
This website and its content may be deemed attorney advertising. Prior results do not predict a similar outcome.
The Mesothelioma Center’s claim as the most trusted resource is based on our more than 150 5-star Google and BBB reviews. Our organization also helps more than half of all mesothelioma patients annually diagnosed.
Your web browser is no longer supported by Microsoft. Update your browser for more security, speed and compatibility.
If you are looking for mesothelioma support, please contact our Patient Advocates at (855) 404-4592
The Mesothelioma Center at Asbestos.com has provided patients and their loved ones the most updated and reliable information on mesothelioma and asbestos exposure since 2006.
Our team of Patient Advocates includes a medical doctor, a registered nurse, health services administrators, veterans, VA-accredited Claims Agents, an oncology patient navigator and hospice care expert. Their combined expertise means we help any mesothelioma patient or loved one through every step of their cancer journey.
More than 30 contributors, including mesothelioma doctors, survivors, health care professionals and other experts, have peer-reviewed our website and written unique research-driven articles to ensure you get the highest-quality medical and health information.
My family has only the highest compliment for the assistance and support that we received from The Mesothelioma Center. This is a staff of compassionate and knowledgeable individuals who respect what your family is experiencing and who go the extra mile to make an unfortunate diagnosis less stressful. Information and assistance were provided by The Mesothelioma Center at no cost to our family.LashawnMesothelioma patient’s daughter


Selby, K. (2026, February 10). p53 Gene Therapy. Asbestos.com. Retrieved March 12, 2026, from https://www.asbestos.com/treatment/tumor-based-p53-therapy/
Selby, Karen. "p53 Gene Therapy." Asbestos.com, 10 Feb 2026, https://www.asbestos.com/treatment/tumor-based-p53-therapy/.
Selby, Karen. "p53 Gene Therapy." Asbestos.com. Last modified February 10, 2026. https://www.asbestos.com/treatment/tumor-based-p53-therapy/.

Dr. Andrea Wolf is the Director of the New York Mesothelioma Program at Mount Sinai in New York City. She focuses on multidisciplinary treatment, clinical research, community outreach and education.
Our fact-checking process begins with a thorough review of all sources to ensure they are high quality. Then we cross-check the facts with original medical or scientific reports published by those sources, or we validate the facts with reputable news organizations, medical and scientific experts and other health experts. Each page includes all sources for full transparency.
Please read our editorial guidelines to learn more about our content creation and review process.
