Guide to Coronavirus for Cancer Patients
Written by Sean Marchese, MS, RN | Edited By Walter Pacheco

Older adults with cancer and chronic illness have the highest risk for developing severe complications from COVID-19. Knowing how to prepare and protect yourself against the virus could make all the difference for you and your family.
Coronavirus: What Cancer Patients Should Know
The new coronavirus is changing life as we know it. The pandemic has quickly spread across the globe, taking national governments and private citizens by surprise.
Its rapid rate of spread and severity of symptoms make the new coronavirus a serious health concern, especially for people with thoracic cancers such as lung cancer and mesothelioma.
Patients diagnosed with these respiratory conditions may be wondering just how dangerous the situation could be for them.
As of Aug. 5, the World Health Organization had reported 18.4 million worldwide COVID-19 cases with 696,147 deaths. The U.S. reported 4,678,610 cases with 154,952 deaths.
What Is Coronavirus?
Coronavirus is the name given to a family of viruses that, when viewed under a microscope, display a spiky ring of proteins around their surface that resemble a crown or corona. SARS-CoV-2 is the virus responsible for coronavirus disease 2019, which is known as COVID-2019. It is the seventh known type of coronavirus to infect humans.
Other types of coronavirus are common among humans and cause mild respiratory symptoms similar to the common cold. The new coronavirus, however, is a strain producing severe flu-like symptoms in most people who come in contact with an infected person.
What Are the Symptoms of Coronavirus?
- Fever
- Tiredness
- Cough
- Sore throat
- Diarrhea
- Breathing troubles
Some patients have reported a loss of smell or taste after infection. However, data on this is lacking, and the effects seem temporary. Other patients are asymptomatic, meaning they show no symptoms but may carry the disease.
Infectious disease researchers currently believe COVID-19 symptoms may not appear for up to two weeks after initial infection; this is known as the incubation period. Getting tested at the first signs of coronavirus infection can protect you from the disease earlier and also prevent the spread to many others.
After getting COVID-19, I noticed my sense of smell and taste were gone. If I was blindfolded, I couldn’t tell you what I was eating.
The Centers for Disease Control and Prevention recommend the regular use of a mask or face covering for the nose and mouth because it is one of the best ways to protect yourself and others from COVID-19.
Respiratory droplets from coughing, sneezing, shouting or simply talking can travel several feet, allowing other people to inhale them.
Studies show that a significant number of individuals infected with COVID-19 may not show symptoms but can still transmit the virus to others. When people in public settings wear masks, they reduce the spread of COVID-19 and can protect those at higher risk of severe illness.

Nose and mouth coverings can be handmade from cloth or purchased from a large number of retailers and independent distributors. They are not a substitute for N95 respirators or surgical masks, which the CDC recommends only for health care workers and first responders.
COVID-19 testing is now available in many locations and is covered by most insurance providers. If you have symptoms of COVID-19 or wish to get tested, the best practice is to contact your health provider first. Your state or local health department’s website can provide the latest information on testing locations.
The U.S. Food and Drug Administration has authorized at-home COVID-19 test kits, which let you collect an oral or nasal sample and send it to a lab for analysis.
If you have symptoms of COVID-19 and do not yet have a confirmed diagnosis, contact your health care provider if symptoms worsen or if you have questions about getting tested.
Remember that if you test negative for COVID-19, it only means you did not have the virus at the time of testing. It does not prevent you from later contracting or spreading the virus.
I already had to cancel one of my appointments…because it’s not worth the risk of me going down there and possibly getting this virus.
How Do I Get Tested for Coronavirus?
- Call ahead and inform your oncologist’s office or emergency department you may have COVID-19. This helps the office protect their staff and other patients.
- Wear a face mask as soon as you begin to exhibit symptoms. Wearing a mask and keeping at least six feet away from others will help prevent the spread of disease.
- Results are available four to six hours after testing. If you test positive, stay home, keep in touch with your doctor and limit contact with other people. If you test negative, it does not mean you won’t get sick later. You should continue following preventative measures.
Most people who are mildly ill with COVID-19 have safely recovered at home. However, cancer patients and other populations with compromised immune systems have a higher risk of severe symptoms.
Because cancer patients, especially those with mesothelioma and lung cancer, have an increased risk of complications from an infection, they should understand the risks of exposure to the disease.
I looked high and low for coronavirus testing sites, but they’re not as easy to find as they say. I had to make an appointment with a doctor to get tested.

Access top mesothelioma cancer centers that have experience treating this rare disease.
Get Help NowWhat Should I Do if I Test Positive?
If you test positive for COVID-19, the best thing to do is stay home and prevent the spread of infection. From home, you’ll be able to track the progression of symptoms and determine whether you should go to the hospital.
According to the CDC, if you experience any of the following emergency warning signs you should seek medical care immediately:
- Trouble breathing
- Constant chest pain or pressure
- Confusion or difficulty concentrating
- Severe tiredness
- Blue or purple discoloration of face or lips
Call 911 or ask a friend or family member to call for you and inform the emergency facility ahead of time that you may have COVID-19.
If you are self-isolating at home with mild symptoms, you should stay well hydrated and get plenty of rest. If you have fever or pain, nonsteroidal anti-inflammatory drugs (NSAIDs), such as Tylenol or ibuprofen, can help relieve symptoms.
The CDC reports there is no evidence suggesting a link between NSAIDs and the worsening of COVID-19. Patients should use NSAIDs, and all other medications, according to the product labels and advice of their health care team.
You can discontinue self-isolation and be around others after both of the following:
- Symptoms are gone or mild 10 days after your positive test date
- You have had 24 hours without fever or fever-reducing medication
If you were severely ill with COVID-19 or had a weakened immune system during the infection, you may require up to 20 days of self-isolation after symptoms first appeared. In all cases, talk with your health care provider to determine when it’s safe to be around other people again.
How Does Coronavirus Affect the Lungs?
As coronavirus attacks the lungs, it causes lung tissue to produce increased fluid and cellular waste known as exudate. This buildup of fluids and matter causes inflammation of vital tissue such as the lining of air sacs and the pleura, which is the tissue surrounding the lungs. In cancer patients, these tissues have likely already been damaged by cancer cells or various treatments.
Damaged tissues in the lungs are more prone to infection as they lack the natural defenses of healthy cells. This allows the development of opportunistic infections, such as pneumonia, which puts patients at risk for septicemia or other complications that can lead to organ failure or death.
By the time the EMTs arrived, my oxygen level was 76% because of COVID-19. They were looking for blood clots. I found out the next morning that I had a partially collapsed lung.
We’ll be updating this page regularly as the situation changes and more resources become available. Below is a list of national information centers providing additional updates and guides on how to prevent the spread of disease and what to do if you’re infected.
Resources With the Latest Updates on COVID-19:
Protecting Cancer Patients from Coronavirus
A COVID-19 vaccine offers the best protection for most cancer patients. Treatment is largely similar to what exists for influenza: Treating the symptoms. According to the Centers for Disease Control and Prevention, the best way to prevent illness is to avoid exposure to the virus.
Coronavirus is thought to spread primarily from person-to-person via respiratory droplets, which are tiny drops of fluid produced when you talk, cough, sneeze or breathe heavily. These droplets can contain the virus and be launched a little more than 3 feet. If any droplets land on another person, or are transmitted by touch, the virus can gain access to the eyes, mouth or nose and begin a new infection.
Due to the nature of coronavirus transmission, social distancing, hand-washing and general hygiene become critically important. Cancer patients can protect themselves from infection by following these CDC-recommended procedures:
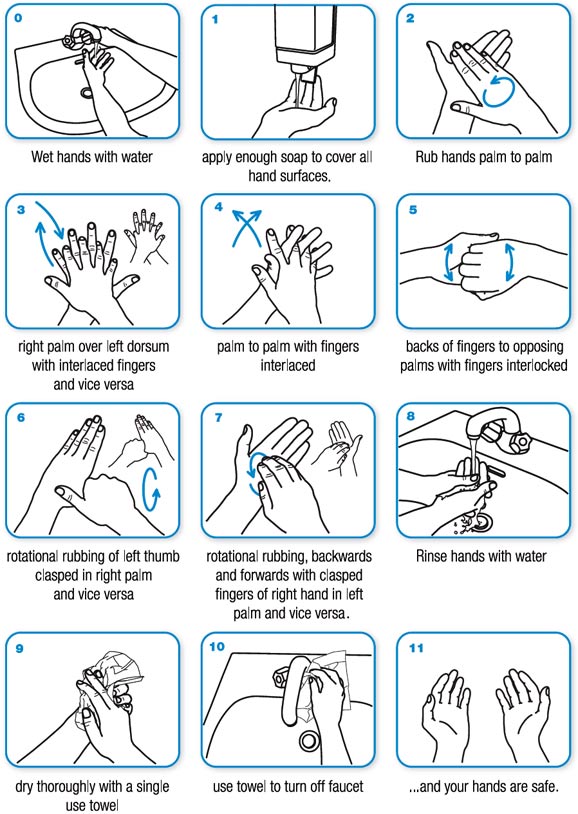
- Wash your hands often with soap and water for at least 20 seconds.
This is especially important after touching someone else, being in a public place, coughing, sneezing or blowing your nose. Avoid touching your face with unwashed hands. If soap and water are not available, use a hand sanitizer that contains at least 60% alcohol to cover all surfaces of your hands and allow them to dry.
- Practice social distancing.
Avoid close contact with people who are sick as well as those who might be sick or who are at higher risk of health complications. Keeping at least 6 feet away from any other people at all times can significantly limit the spread of COVID-19. This includes staying home when you are sick or wearing a face mask if you must leave the house. You do not need to wear a face mask if you are not sick.
- Keep surfaces clean and disinfected.
Doorknobs, sinks, countertops, tables, remote controls, switches, keyboards, phones and any other frequently touched surfaces in your home should be cleaned and wiped with household disinfectants.
World Health Organization, 2009
Social Distancing Considerations for Cancer Patients and Caregivers
If you are a cancer patient or caregiver accustomed to being around someone else almost every day, it may be difficult to adjust to social distancing. However, there are many ways to work around the challenges of long-distance caregiving.
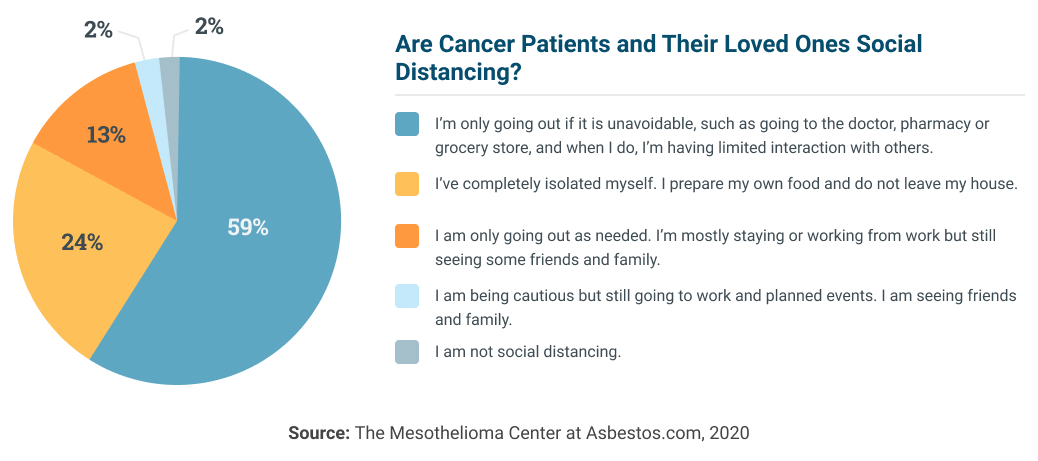
First and foremost, caregivers should be following the CDC and World Health Organization guidelines and policies. Adults 60 and older with preexisting medical conditions are at the highest risk for coronavirus complications. Washing your hands, avoiding crowds and disinfecting surfaces could save lives.
The fatality rate across all coronavirus patients with a history of cancer was 5.6%.
Limiting in-person visits will lower transmission risk of COVID-19. But it may be tough for older patients who are used to spending time with friends and family. However, social distancing does not need to lead to isolation.
Caregivers can help their loved ones access technology that will allow them to reach out and continue connecting with the people they care about. Cancer patients who need to speak with their attorneys for new or ongoing litigation may also use teleconferencing tools to communicate with their lawyers.
TIPS TO EASE SOCIAL DISTANCING PROBLEMS
- Learn how to make video calls with family members using a phone, tablet or computer.
- Set up accessibility options, such as increased font size and volume, on devices.
- Register for grocery deliveries through Amazon or meal kit deliveries, such as BlueApron or HelloFresh, instead of eating out (make sure to transfer food out of containers and wash thoroughly to prevent infection).
- Use TaskRabbit to hire someone for household chores, repairs or miscellaneous tasks. TaskRabbit workers continue providing service during COVID-19.
- For nonessential doctor visits or routine health care, use a telehealth service to connect over video with a provider who can refill prescriptions or monitor your health.
Easing COVID-19 Worries of Caregivers and Family
Caregivers, family members and loved ones may be wondering how best to keep themselves and others safe during shelter-in-place orders. Depending on your situation, it may be best to limit interaction with your patient, prepare a stock of supplies or alter travel plans.
Adapting your role as a caregiver to be safe and responsible while still providing care is the best thing you can do to keep yourself and your loved ones safe during the pandemic.
What happens if my patient or I must be quarantined?
Caregivers and family members can follow these guidelines for effective self-quarantine:
- Stay at home
- Do not permit visitors
- Stay 6 feet apart from others
- Keep surfaces and objects disinfected
- Do not share utensils, towels or clothes
Elective cancer surgeries were scheduled to resume April 26, 2020. Cancers such as lung cancer are more treatable through surgery than other methods and waiting more than a month poses more risk than benefit. However, the risk of infection becomes significantly greater after surgery, making effective self-quarantine that much more important.
If you are a caregiver, consult with your patient’s physician or treatment center regarding safety measures you should take before entering your patient’s home or providing care.
FEMA.gov has a list of emergency management agencies by state that you can contact to find out more details regarding your local shelter-in-place order.
How should I prepare the home for a quarantined cancer patient?
If you have a loved one who could be at risk for COVID-19, take the opportunity to prepare their house to prevent transmission of disease. Use disinfecting wipes that are 60% alcohol to clean frequently used surfaces and electronics.
If you are using an EPA-registered household disinfectant, check the label to determine if the product needs to be left wet on surfaces or if you should be using gloves or a face mask.
Consider creating a safe zone near the front door or other entrances. Block off an area with tape and set up a table with hand sanitizer and a trash can. This way, anyone who must come to the house can remove any possibly infected items from their person and sanitize their hands before entering.
If there are multiple people sharing a home and one of them is sick, choose a room in the house that can be used to separate the sick household member. If possible, allocate a bathroom for only the sick person to use. Clean the sick person’s area only when needed in order to limit your exposure.
To clean soft surfaces, use soap and water or launder according to the manufacturer’s recommendations. Use the warmest possible setting on your washing machine and ensure items are completely dried.

Wear gloves if you are doing laundry for a sick family member. It is not required to separate their laundry from anyone else’s items. However, avoid shaking dirty laundry and disinfect clothes hampers after emptying. When finished, remove gloves and wash hands immediately.
A caregiver can provide personal cleaning supplies, such as paper towels, toilet paper and cleaning solution, to the sick household member. If they are able, the sick person can clean their own space.
What should my caregiver or family member role be during this period?
As a caregiver or family member of someone at risk for COVID-19, your first priority should be your own safety. If you become ill, you will be unable to provide care for those who require it. Limit contact with strangers through social distancing and avoid any public places if possible. Follow CDC guidelines for hand-washing and do not reuse face masks or gloves.
As you work to protect your loved ones from becoming sick, organize their environment with a stock of the following cleaning and health supplies:
- Face masks
- Disposable gloves
- Disposable paper towels
- Tissues
- Waste bins with disposable liners
- Cleaners and disinfectant solutions
- Hand soap and sanitizer (at least 60% alcohol)
- Fever-reducing medication (such as acetaminophen or ibuprofen)
- Thermometer
Your role as a caregiver during this time should also be to provide support and stability. Recommend to your patient or family member they avoid watching the news if it causes them anxiety or concern. Reassure them you will always be available by phone or video call.
Continue to remain in contact with your patient’s physician or treatment center for updates regarding their schedule and availability.
As a caregiver, you have the opportunity to be a pillar of security and comfort for those who need it most. Preparing a safe environment and providing the right resources will ensure those with the highest risk of infection will still be protected.
Should I travel with the patient?
The CDC recommends that people at higher risk for COVID-19 complications avoid all cruise travel and nonessential air travel. Crowded settings, such as airports, may increase your patient’s exposure, and the rate of spread may differ between your origin and destination.
If you are planning domestic travel, consider if it is worth the risk of exposing your patient to the virus. For essential travel, such as to a distant treatment center, make sure that during the trip your patient is separated from other people, wearing a face mask and following strict hygiene precautions.
It is best practice to book a separate adjoining hotel room for the sick patient. If the patient requires more careful observation or frequent medical care, try to book a large room with beds that are farther apart. Assume all surfaces in your hotel room are potentially contaminated and inquire with the staff on what steps they are taking to prevent spread of transmission.
Where can I get financial help during COVID-19 pandemic for my loved one diagnosed with lung cancer?
The LUNGevity Foundation created Breathe Easier: An Emergency Response Fund, which provides $500 grants to eligible lung cancer patients and their families during the coronavirus crisis.
Patients can call LUNGevity’s help line at 844-360-5864 to learn about eligibility requirements.
How Should I Handle a Patient’s Legal Claims While Practicing Social Distancing?
Top mesothelioma lawyers across the United States are taking measures to ensure clients can continue to file asbestos claims within the statutes of limitations. Lawyers have transitioned much of the legal process to virtual methods such as video calls and electronic signatures.
National law firms are still utilizing in-person meetings for patients who prefer face-to-face communication. Mesothelioma lawyers protect their clients’ health by wearing masks and gloves, maintaining an appropriate distance and heavily sanitizing their materials.
Additional Legal Resources
How are end-of-life and palliative care planning affected by COVID-19? What about funerals?
More than half of cancer patients and their caregivers have expressed worry about pandemic factors affecting palliative care and end-of-life decisions.
The pandemic has forced many physicians to begin broaching the topic of advance care planning earlier in a patient’s treatment course. This shift has helped increase the likelihood of patients receiving hospice care and allows time to follow patient requests more appropriately.
As more individuals face the possibility of contracting COVID-19, advance care planning is gaining traction as a standardized part of a patient’s treatment plan. Addressing end-of-life wishes provides a sense of self-advocacy for the patient during a time when many other factors may be out of their control.
Hospice care and palliative care options remain available during the COVID-19 pandemic, especially as hospitals and health care systems recoup resources after the initial surges of COVID-19. Cancer patients and caregivers should have this discussion with their providers early to maximize the options available.
Funeral planning can be especially difficult during this time, and the additional burden of grief after the loss of a loved one can be overwhelming. Decisions on how to safely conduct a funeral service may be difficult, but funeral directors and community leaders are working with families to address these concerns.
The CDC recommends the following guidelines for safe funeral gatherings:
- Attendees should wear masks during the service, primarily if social distancing cannot be maintained
- Gatherings in outdoor or well-ventilated areas lower the risk of transmission
- Avoid shared objects such as religious books, pamphlets or other communal items
- Reduce activities such as singing or chanting, which can increase the number of viral particles in the air
Additional CDC guidelines are detailed extensively here and include limiting attendees to close family and friends, altering traditional rituals and avoiding contact with the deceased’s body.
Impact of COVID-19 on Lung Cancer and Mesothelioma Patients
Older adults with thoracic cancers and other respiratory illnesses are the populations most at risk from COVID-19. The coronavirus is a respiratory disease and presents unique challenges and complications for patients already struggling with respiratory symptoms and distress.
There are a number of preexisting conditions that make the fight against COVID-19 tougher for many patients. Lung cancer, mesothelioma, head and neck cancer or chronic obstructive pulmonary disease all present the opportunity for dangerous complications with coronavirus.
Mesothelioma patients may also have difficulty identifying worsening symptoms of their cancer versus signs of a COVID-19 infection. This similarity makes COVID-19 testing especially crucial for patients with mesothelioma and lung cancer.
Having coronavirus felt like an elephant was sitting on my chest. As I breathed, it was like my chest was burning. When I would talk, I would have shortness of breath and become lightheaded, and my heart would race.
Lung cancer patients are at high risk of coronavirus complications, according to a JAMA Oncology study published March 25, 2020. Among 228 patients with non-small cell lung cancer at a facility in Wuhan, China, seven patients were diagnosed with COVID-19 pneumonia.
The study reported that hospital admission and frequent hospital visits were the greatest potential risk factors for infection. Further evidence also indicates that smoking history and COPD are independent risk factors in severe COVID-19 cases.
Symptoms of Chronic Illness vs. Infection
People with lung cancer have a 50% to 70% chance to develop serious lung infections, such as pneumonia, during their illness. Cancer patients and other people with weakened immune systems should be aware of the differences between symptoms of an infection versus those of a chronic respiratory illness.
The symptoms below appear in both respiratory infections and thoracic cancers, however there are some key distinctions:
- Cough: Coughing will be more persistent in thoracic cancers, lasting several weeks and getting progressively worse.
- Shortness of breath: People with pneumonia will have short and severe bouts of breathlessness that may progress rapidly. In lung cancer and mesothelioma patients, shortness of breath is typically more persistent and regular.
- Fatigue: Pneumonia may cause tiredness, but it is usually not as severe as fatigue in cancer patients.
- Loss of appetite: Anorexia, or decreased appetite, is more common in cancer than in pneumonia. This is the primary cause of weight loss in mesothelioma and lung cancer patients and can be mitigated by a healthy diet.
- Phlegm, wheezing or chest pain: These are similar in presentation in thoracic cancers and pneumonia. If these symptoms worsen, contact your physician for further testing.
Importance of Cancer Diagnosis
Patients experiencing symptoms of COVID-19 should not discount the possibility of a cancer diagnosis. Many signs of infection, such as cough, fever and fatigue, are also indicators of thoracic cancers such as mesothelioma and lung cancer.
Patients with a history of smoking or asbestos exposure have a significantly higher risk of developing cancer and should speak with their doctor about cancer screening during this time. A COVID-19 infection presents the possibility of severe complications for patients with thoracic cancer.
The medical journal Science recently published a study that reported a steep drop in cancer diagnoses since the start of the pandemic. However, the report clarifies that the actual incidence of cancer has not decreased. Patients have had fewer routine cancer screenings because of the fear of contracting coronavirus in health care settings.
Some doctors have also paused routine screenings and annual exams in favor of handling the onslaught of COVID-19 cases. However, the delay of accurate cancer diagnoses may translate to more untreated cancer cases over the next decade. The report projects an increase in several thousand cases that doctors may not diagnose until the later stages.
Patients need to speak with their physicians about their cancer risk factors, such as smoking and asbestos exposure. COVID-19 symptoms and screening should take priority, but not replace a necessary cancer exam outright. Delaying a cancer diagnosis could mean a difference in the availability of lifesaving procedures.
Risk of Misdiagnosis
Because of the overlap in symptoms between respiratory infections and thoracic cancers, the risk of misdiagnosis is much higher during the COVID-19 pandemic. Currently, understaffed and undersupplied medical providers worsen this situation.
A patient who has a cough and fever may test negative for COVID-19 and not receive further screening for thoracic cancers such as mesothelioma and other asbestos-related cancers. Similarly, a cancer patient who experiences chronic cough and respiratory distress may find it more difficult to identify the symptoms of COVID-19 and know when to get tested.
Common symptoms that occur in thoracic cancers and not in respiratory infections include:
- Vocal changes such as hoarseness
- Unexplained weight loss
- Swelling in the face or neck
- Difficulty swallowing
- Joint pain in the shoulders or neck
- Changes in the shape or color of fingertips and nail beds
Dangers of Pneumonia in Cancer Patients
Infections are the second most common cause of death in people with lung cancer. Without a diagnosis and proper treatment, complications can lead to permanent damage and possibly death.
As pneumonia develops from severe coronavirus infections, it can cause severe damage to mesothelioma and lung cancer patients who have undergone aggressive treatment or only have one lung remaining.
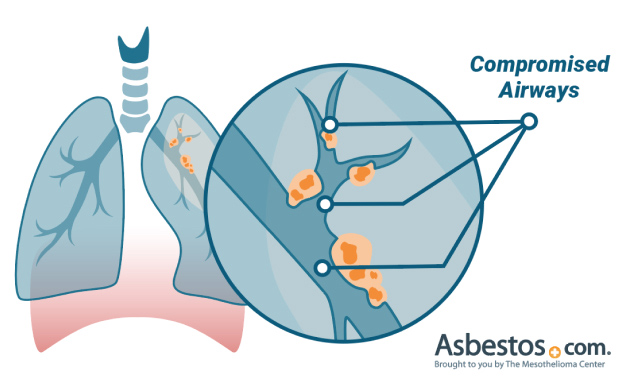
Tumor mass in the lungs or chest cavity takes oxygen and essential nutrients away from healthy cells. The cancerous tumors cause a decrease in normal lung cell function. Not only does this increase the risk of infection as the airway becomes compromised, but it also prevents the ability to clear the infection.
A person with metastatic lung cancer who develops pneumonia has a 5-year survival rate of less than 5%. In contrast, the survival rate of people infected with pneumonia without lung cancer is more than 95%.
Pleural mesothelioma patients are at higher risk of chest pain with pneumonia because the nerve fibers that sense pain are located in the pleura. This pain is usually sharp when inhaling a deep breath and is called pleuritic pain or pleurisy.
How Can Cancer Patients Remain Prepared?
Stockpiling of household necessities continues to be a concern as many retailers run short on supplies. Unfortunately, cancer patients also require medical supplies and equipment, such as oxygen, that may be hard to obtain or in high demand.
Contact your regular supplier of oxygen or medical supplies and find out if they are still delivering to homes during the pandemic. Many suppliers have mobilized to provide support in wider areas that require assistance. If your regular supplier is unavailable, contact your health care provider for a list of services that provide the equipment you need.
Practicing social distancing does not mean you won’t get the deliveries you need. Call ahead to your delivery services and inform them if you or anyone at home is possibly sick. Many delivery companies are prepared during this time to offer contactless delivery or drop-off and prevent contamination of your home or their employees.
Social distancing may also provide challenges for cancer patients who have recently undergone surgery and are more susceptible to infection. People who are still recovering from surgery and require caregiving at home should continue to follow the CDC and WHO policies and procedures for preventing infection and decreasing risk of transmission.
If you are at risk for infection, it is essential to keep open communication with anyone who may come into your home to determine if they have been potentially exposed to the coronavirus.
Additional Mesothelioma Resources
Cancer Treatment and COVID-19
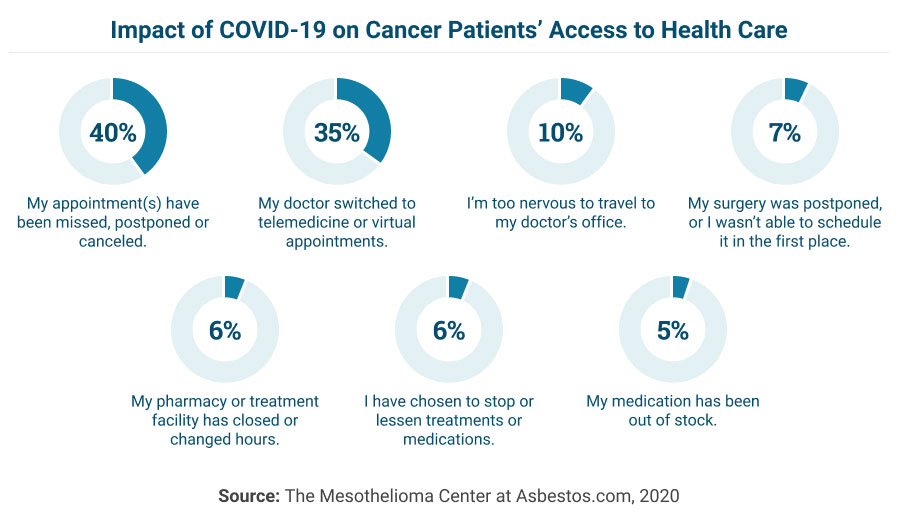
Because of the overwhelming number of people seeking medical care and the burden that COVID-19 is placing on health care providers, it may be more difficult to access your regular cancer treatments during this time.
Many patients may be experiencing anxiety or reluctance to visit their physician’s offices and expose themselves to others who may be infected. For this reason, it’s important to call ahead wherever you plan to be seen. The office can make accommodations for you and their staff, so you are only in proximity with essential staff.
Appointment changes, treatment disruptions and pharmacy availability are also considerations that may impact your regular schedule of health care. If you have not received updates, make sure to contact your pharmacy and health care providers and inquire about any altered business hours or unavailable services.
In addition to medications being out of stock, planned surgeries may also be postponed, reducing unnecessary infection risks for vulnerable populations. Cancer patients may feel worry or anxiety as they are left waiting for the treatment required to continue treating their cancer.
These feelings are valid but can lead to irritability or fatigue. Keep clear and organized documentation of your health and symptoms to ensure you’re prepared for any disruptions in your regular care.
Have medication lists, medical history, test results and similar medical documents available in case you are required to continue treatment at an alternative site.
Treatment While Diagnosed with COVID-19
An April study in the American Journal of Clinical Oncology reported that cancer treatment within 14 days of a COVID-19 diagnosis was a risk factor for developing the following severe events:
- Acute respiratory distress syndrome
- Septic shock
- Heart attack
Patients with severe disease will likely require hospitalization. Inpatient management currently focuses on treating the most common complications of the disease, including pneumonia, respiratory and cardiac failure, kidney injury and blood clots.
Some hospitals are employing a technique called “proning” to improve oxygenation levels in patients with respiratory issues. Proning involves having the patient lay face down while breathing. This position reduces weight on the organs and improves the lungs’ ability to expand when taking a breath.
Proning and breathing exercises, such as the use of an incentive spirometer, are methods of improving oxygen levels in patients. These practices are vital to preventing respiratory complications, which may require the use of a ventilator.
Complications of a COVID-19 infection can prolong the time it takes to resume cancer treatment after hospitalization. Recovery might take several weeks, and your oncologist may recommend waiting before continuing chemotherapy or other anti-cancer care.
Patients recovering from COVID-19 should discuss the risk of cancer recurrence with their health care team, as these factors are different for every patient.
Status of Cancer Clinical Trials During Pandemic
Treatment centers are beginning to resume mesothelioma and cancer clinical research trials across the United States. Thanks to the National Cancer Institute’s measures, health care providers have been able to make adjustments to clinical trial visits and study activities in order to keep patients safe.
The NCI has worked with institutional review boards and trial sponsors to provide increased flexibility for the timing of patient tests and assessments. In most cases, your oncologist will handle clinical research trial activities as they would regular care, and treatment schedules will be at the discretion of your provider.
Any nonessential appointments with research staff, such as questionnaires or research-specific blood work, may be postponed or canceled.
The pandemic may impact the availability of experimental medication through clinical trials, depending on location. Shipments of drugs may be delayed or drugs might simply be unavailable. The research staff will inform you and your physician of any potential interruptions in treatment during this time and provide you with options.
If your medication becomes unavailable or a treatment delay will be too long, your physician will temporarily switch you to conventional treatment until the clinical trial resumes operation.
Additional Clinical Trial Resources
Mesothelioma patients undergoing treatment at specialized facilities for clinical research trials or targeted therapies may have additional concerns. Many treatment centers are returning to standard capacity and operations, but it’s essential to call ahead and prepare for any changes to your next visit.
There are additional steps you can take to stay on track with your planned treatment schedule. Below are some frequently asked questions regarding how the coronavirus pandemic may affect your cancer treatment.
I receive cancer treatment at a medical facility. Should I continue my cancer treatment? Should I find a way to get care at home?
The answers to these questions will come from the medical facility where you currently receive treatment. If you have not heard updates from them regarding changes in schedule or operations during the COVID-19 pandemic, contact them to discuss if your treatment will be affected.
There is currently no evidence that suggests you should stop taking any of your regular prescription medication.
If you have routine scans or tests scheduled, consult your physician if it’s worth delaying these appointments, especially if you are clinically stable and without symptoms.
If your treatment center is moving to limited or discontinued availability, follow up with them and find out if they can provide a list of alternative resources or places to receive treatment.
You can also contact your insurance provider for a list of facilities where you can temporarily access your treatment. They may also be able to inform you of any home care options available. It’s most important to speak with your oncologist, however, to ensure you are continuing the therapy recommended for your condition.
If alternative treatment centers or services are needed, call ahead to any transportation companies you may need to use. Make sure these companies are equipped to provide you with service on the dates and to the locations you require.
Whether you will be seeking care at a different facility or registering for home care for the first time, continue to follow the recommended guidelines for hand hygiene, disinfecting your home and social distancing.
What can I expect at my treatment facility?
As the situation unfolds and more people seek medical care, the quality of services and options you may be accustomed to at your treatment center could be limited.
- Visiting hours for friends and families may be limited or restricted.
- There may be increased screening procedures and precautions.
- You may expect delays in treatment or scheduling appointments.
- Medical staff may be reduced, increasing wait times and limiting services.
- Supplies, such as medical masks and oxygen, may be limited.
- You may be screened at the door to determine if you are symptomatic or have a fever.
- Many patients are being asked to wait in their car before being called to come inside.
- Patients are restricted from gathering or waiting in lobbies and access to services, such as water or coffee machines, may be limited.
It is important to maintain contact with your cancer care team to determine the best course of action for you.
What precautions do patients and caregivers need to take?
Limiting contact with other people is the primary way to reduce infection risk. Cancer patients should refrain from close proximity to anyone who may be sick or could have recently come in contact with someone who has coronavirus.
Proper precautions must be taken if physical contact is required for caregiving. Frequent hand-washing is important for caregivers and patients and should be done with soap and water for at least 20 seconds. Continue to disinfect any frequently touched objects or surfaces with household cleaning solutions.
A replacement caregiver should be appointed if the primary caregiver is experiencing any symptoms or has been exposed to anyone ill. If an alternative caregiver is not available, contact your physician about making arrangements for a visit or possible home health care services.
In most cases, your oncologist will handle clinical research trial activities as they would regular care and treatment schedules will be at the discretion of your provider. Any nonessential appointments with research staff, such as questionnaires or research-specific blood work, may be postponed or canceled.
Experimental medication available through clinical trials may be affected. As more personnel enter quarantine, shipments of medications may be delayed or simply unavailable. The research staff will inform you and your physician of any potential interruptions in treatment during this time and provide you with options.
If your medication becomes unavailable or a treatment delay would be too long, it is likely your physician will temporarily switch you to conventional treatment until the clinical trial resumes operation.
What should I know while taking chemotherapy?
Chemotherapy compromises the immune system and increases the risk of infection. For this reason, it is essential to follow strict hand-washing and social distancing guidelines from the CDC and WHO.
If you are actively receiving chemotherapy during the COVID-19 pandemic, speak with your treatment center staff and physicians to ensure they are taking all precautions necessary to protect you from transmission. Ask about their policy for keeping sick employees home and whether they have the proper protective equipment to serve you without risking your health.
Patients receiving chemotherapy should stay at home at all times except when treatment is required. Delivery and other online services can assist with groceries and other tasks when needed. Staying home during this period is crucial to preventing infection.
Will my doctor stop my lung cancer or mesothelioma treatment if I get coronavirus?
In most cases, your physician or mesothelioma specialist will withhold chemotherapy or other cancer treatments to give your body the best chance at fighting a coronavirus infection.
Treatments such as radiation therapy may take over six weeks to complete while some chemotherapy patients receive treatment continuously to prevent cancer recurrence. These prolonged treatment periods offer windows where it is generally safe to postpone or withhold treatment for one to two months.
Patients undergoing radiation therapy alone are likely to have their treatment schedule adjusted to allow for fewer visits through hypofractionation. This process allows radiation oncologists to deliver the same amount of radiation across fewer treatments and reduce the risk of infection with frequent visits to the treatment center. This technique isn’t recommended for patients undergoing chemoradiation.
Chemotherapy, radiation therapy and surgery are some cancer treatments that make it more difficult for your body to fight infection. Because the risks of severe complications, such as pneumonia and septicemia, are so high with coronavirus, treatment will likely be withheld so your immune system can fight back.
Your oncologist may also recommend you visit a specialist during this time (if you’re not seeing one already) in order to ensure you have the most options available if you become infected.
Are there treatments or a cure for coronavirus?
There is presently no antiviral treatment specific to COVID-19. However, a phase 3 trial known as ACTIVE-3 is currently ongoing at select hospitals around the world. The trial is testing experimental COVID-19 monoclonal antibody treatments.
Many studies are underway to develop a vaccine for COVID-19. The National Institutes of Health has partnered with biotechnology company Moderna to produce manufactured viral proteins that can lead to a vaccine.
Moderna published a report in July 2020 on a promising investigational vaccine, mRNA-1273, which has been tolerated well and is prompting antibodies in trial participants. Plans for a phase 3 trial are underway.
Scientists are also testing plasma-derived therapy as a potential treatment for COVID-19. Takeda Pharmaceutical Company is experimenting with collecting antibodies from patients who have recovered from the virus and injecting them into newly infected patients, signaling the body to produce an immune response. So far, anecdotal results have been positive.
Questions to Ask Your Doctor About COVID-19
More than half of cancer patients have not spoken with their doctor about coronavirus, according to a 2020 survey by The Mesothelioma Center. Getting the information you need to get tested, continue treatment and keep your loved ones safe is critical, and much of that communication will come from your medical team.
You can ask your physician the following questions about coronavirus:
- Should I go to the hospital if I have certain symptoms?
- How do I get tested?
- Am I at a higher risk of getting coronavirus or COVID-19?
- Should I be seeing another type of doctor or specialist for now?
- How can I prevent getting coronavirus in my situation?
- What will happen if I’m positive for coronavirus?
- What does it mean if I test negative and still feel sick?
- How can I receive care at home?
- Is telehealth available for my care, or can I be seen by a doctor over the internet?
- How should I alter my exercise and activity routines?
- Will I be able to continue treatments?
- Should I continue treatments, or is it safer to wait?
- What happens if I miss a treatment?
- Can I go to work? Can my caregiver go to work?
- Is there a list of alternative treatment centers I can go to?
- Does my treatment lower my immune system?
- What kind of protective equipment should I have at home?
- What symptoms should I be concerned about?
- How can I get essential food and medical supplies delivered to my home?
- Who can I let into my home?
- What should I know while I’m getting chemotherapy?
- Are there any federal or government policies that could affect my care?
Is it safe for cancer patients to go out if my state is reopening? What are the criteria for reopening?
The White House and CDC have published “Guidelines for Opening Up America Again.” Phased reopenings are occurring on a state-by-state basis, and some criteria may be altered based on local circumstances.
In general, regional gating criteria to reopen businesses involves the following:
- Downward trajectory of flu-like illnesses and documented cases of COVID-19
- Ability of hospitals to treat all patients without crisis care
- Robust testing program in place for at-risk health care workers
The goal of reopening in phases is to mitigate outbreaks, protecting the health and safety of workers and the general public. Health care systems must have the resources necessary to handle surges in cases for both noncritical and ICU patients.
Cancer patients should continue to practice good hygiene and social distancing, even after their state reopens. People who feel sick should not go to work or gather in public spaces.
Recommended guidelines for individuals in areas approved for Phase One reopening include:
- Vulnerable individuals should continue to shelter in place
- Avoid social gatherings of 10 or more where physical distancing is not an option
- Minimize nonessential travel and follow CDC guidelines for isolation during travel
Phase Two criteria are similar to the above recommendations, but guidelines restrict social gatherings of 50 or more people in areas where appropriate distancing may not be practical. Additionally, nonessential travel is permitted.
For states where Phase Three reopening guidelines are in place, the CDC recommends the following:
- High-risk individuals can resume public interactions but should continue to observe social distancing and precautionary measures
- People at low risk should minimize the time spent in crowded environments
Contact your department of health for the latest information on your region’s reopening status.
Cancer patients should consider themselves at high risk for contracting COVID-19 and practice extreme caution when engaging in social activities or visiting businesses and other establishments.
Additional Treatment Resources
Effect of COVID-19 on Veterans
For veterans with disabilities and service-related illnesses such as mesothelioma, most VA activities have resumed normal operations since the start of the COVID-19 pandemic. Many VA facilities are open and continue to assist veterans with filing benefits claims and seeking medical care.
Veterans who suspect they may have symptoms of COVID-19 can receive clinical screenings at any VA facility through the VA’s emergency management coordination cell. VA facilities are offering diagnostic testing for veterans enrolled in VA health care.
To receive testing for COVID-19 at a VA facility, send a message to your VA health care provider, schedule an appointment online or call your provider’s phone number. The VA recommends that only those with an appointment go in person to a VA health care facility.
Veterans who have lost their jobs or have a reduced income may qualify for enhanced eligibility status based on their income. Enhanced eligibility means the VA will place you at a higher priority for health care and other benefits.
The VA is now processing benefits without delay, including disability, pension and education payments. Some regional offices may be closed for in-person claim filing, but claims can still be submitted by mail, online or with help from a VA-accredited claims agent.
For veterans with service-related disabilities such as mesothelioma, the VA is also conducting claim exams via phone or video through telehealth services. VA-accredited claims agents can help you process claims through video or phone calls and prevent any delay in benefits.
Staying Healthy During Coronavirus Pandemic
The coronavirus pandemic is a frightening time for everyone. Many things may seem uncertain, and the country is scrambling to figure out a new way of life.
However, in this time of crisis, we can choose to continue living healthy and meaningful lives. Strength and support from our friends and families will guide us through this period in history, and we can grow better from it.
There are more opportunities now than before to focus on making healthy choices and discover better ways of living. Concentrating your energy on a positive outlook will give you the resolve you need to persevere.
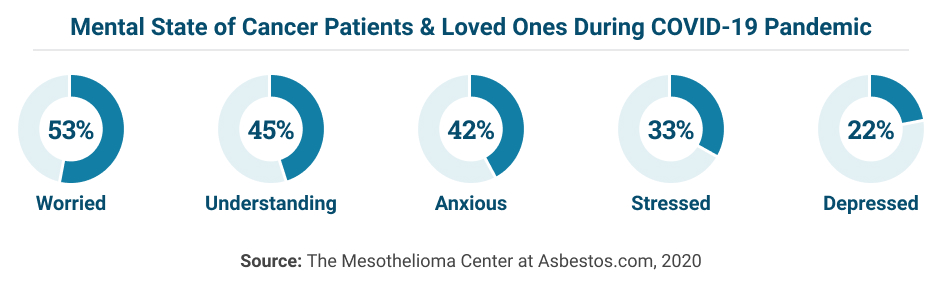
Eating Healthy
Taking steps to care for your emotional and mental well-being during the coronavirus pandemic can start with eating the right foods. Cancer survivors who develop healthy eating habits are proven to have a better quality of life and better treatment outcomes.

The American Cancer Society recommends these steps to improve nutrition:
- Eating a variety of fruits and vegetables
- Fewer processed meats and red meats
- Fewer sweets and drinking less alcohol
- Substituting with whole grains and brown rice
Being at home more is a good time to experiment with cooking and trying new recipes. If you have the need to snack throughout the day, consider fresh fruit or mixed nuts. When ordering delivery while staying at home, look up the menu beforehand for healthy options.
Staying Active at Home
According to the American Cancer Society, adults should aim to get at least 150 minutes of moderate-intensity, or 75 minutes of high-intensity, activity each week.
Moderate-intensity activity includes exercise such as a brisk walk, which should make you breathe hard. Vigorous activities increase your heart rate substantially and will make you sweat as you use large muscle groups.
If you are working from home, consider walking around while you take phone calls or respond to emails. When sitting down for work, you can do leg lifts or bicep curls. Set an alarm to take a break every hour for a quick walk or stretches.

Other ideas for activity at home:
Building Support During COVID-19
All cancer patients should be aware of the benefits of a healthy lifestyle, including proper nutrition, adequate exercise and emotional well-being. Building a support network may be difficult during the COVID-19 pandemic, but the right amount of self-care can help you overcome any obstacles.
Staying at home with cancer during the coronavirus outbreak can produce many troubling feelings and reactions. Staying positive is important for your overall health. The NIH provides some tips on strengthening your emotional wellness:
- Keep a good outlook on life, reinforce healthy habits and reflect on positive experiences
- Stay relaxed and get enough sleep at night
- Read new books, try a new recipe or discover a new hobby
- Keep frequent contact with family and friends through text, video calls or e-cards
- Look for ways to laugh or have fun such as playing an online game or watching movies
- Focus on spiritual enrichment, journaling, making art, enjoying nature or meditating
- Join an online support group or discussion community
These are difficult times, and while COVID-19 is a serious concern, there is no reason to panic. Following the recommended guidelines and remaining in contact with your medical team are the best measures you can take to weather the storm. You can also help educate friends and family on best practices for keeping others safe.
Many physicians and health care providers are facing extreme stress during this time because of the overwhelming anxiety of their patients and the difficult decisions they face in delaying care. If you have friends or family who work in health care, consider reaching out and offering your gratitude, love and support.
Anxiety and fear are normal during this time; allowing yourself to develop these feelings can help you cope with them. Limiting your news intake, staying connected with loved ones and maintaining daily routines will make it possible for you to overcome fear and feel positive about the future.
Additional Support Resources
The Mesothelioma Center at Asbestos.com has an active and enriching community built by our Patient Advocates who are always happy to answer your questions. Regardless of how long the coronavirus pandemic lasts, always remember there is hope and support all around you.
I am trying to stay positive, but this isolation and inability to see doctors in a time like this with my situation is very scary and mentally taking a toll.
Additional COVID-19 Information and Resources
- Coronavirus: What Mesothelioma Cancer Patients Should Know
- Virtual Mesothelioma Treatment Options During COVID-19
- Mesothelioma Clinical Trials Resume After COVID-19 Disruption
- Mesothelioma Patients, Caregivers Can Manage Coronavirus Anxiety
- Safely Handling Mesothelioma Claims Amid Coronavirus Scare
- COVID-19 Closures Accelerate Asbestos Abatement in Schools
- Libby Protects Those with Asbestos Disease from COVID-19











