
Connect with top-rated doctors specializing in mesothelioma treatment, who will personalize treatment options based on your diagnosis.
Find Your DoctorIpilimumab, marketed under the brand name Yervoy and manufactured by pharmaceutical giant Bristol-Myers Squibb, is an immunotherapy treatment for late-stage melanoma. Clinical trials are testing Yervoy in people with mesothelioma, and early results are promising.
Yervoy is the brand name of ipilimumab, an immunotherapy drug approved to treat mesothelioma. The pharmaceutical company Bristol Myers Squibb manufactures it.
Since its approval by the U.S. Food and Drug Administration in 2020, doctors have begun using Yervoy to treat adult patients with unresectable malignant pleural mesothelioma. Yervoy is currently only approved as a first-line mesothelioma treatment combined with nivolumab (Opdivo).
Yervoy is a monoclonal antibody and part of the immune checkpoint inhibitor drug class. It binds to the CTLA-4 protein to prevent immune suppression. Doctors may prescribe Yervoy every six weeks with nivolumab every three weeks.
Clinical trial results across many U.S. cancer centers have been promising. They show that Yervoy is effective against mesothelioma when combined with Opdivo.
The CheckMate 743 clinical trial paved the way for Opdivo and Yervoy’s FDA approval to treat pleural mesothelioma. In April 2022, the study posted an update on its 605 pleural mesothelioma patients. Those treated with Opdivo and Yervoy had a median survival of 18.1 months compared to 14.1 months for chemotherapy alone.
In January 2022, the National Cancer Institute opened a clinical trial testing Yervoy with LMB-100. The latter is a protein engineered to kill cancer cells. The NCI opened the phase I trial to pleural and peritoneal mesothelioma patients.
The University of Chicago Medicine Comprehensive Cancer Center began enrolling patients in 2020 for a multimodal therapy trial. It is examining the effects of Opdivo and Yervoy when added to peritoneal mesothelioma surgery.
In June 2017, French researchers announced the results of phase II clinical trials of Yervoy and Opdivo. All participants in the study had inoperable mesothelioma that had returned following one or two lines of chemotherapy with pemetrexed and cisplatin or carboplatin.
Approximately 50% of the 54 participants treated with the immunotherapy drug combination responded well. Tumors shrunk in nearly 26% of patients, and another 24% of patients showed no new tumor growth.
Results such as these are remarkable in the treatment of recurring mesothelioma. This cancer is resistant to treatment. These results have researchers excited about what future studies may hold for mesothelioma.

Connect with top-rated doctors specializing in mesothelioma treatment, who will personalize treatment options based on your diagnosis.
Find Your DoctorA 2020 phase II clinical research trial sponsored by Bristol Myers Squibb is testing the progression-free survival benefit of the UV1 cancer vaccine with nivolumab and ipilimumab in malignant pleural mesothelioma patients. Eligible patients must have disease progression after standard chemotherapy. Preliminary results are expected in 2022.
In 2017, researchers launched a phase II study testing the novel antibody INCAGN01867 with Opdivo, Yervoy and Opdivo plus Yervoy. The trial is targeting patients with advanced or metastatic malignancies, including mesothelioma. It was completed in November 2021, but early results are unavailable.
In 2016, researchers in the Netherlands launched a landmark trial called INITIATE. It studied ipilimumab and nivolumab as a second-line treatment for pleural mesothelioma.
The phase II study followed 33 patients with unresectable mesothelioma. They had disease progression following platinum-based chemotherapy. Researchers found that combination therapy promoted T cell proliferation. Treatment with Opdivo alone did not.
MAPS2 studied the effects of Opdivo and Yervoy as second-line therapy for pleural mesothelioma. MAPS2 launched in 2016 and enrolled 125 patients. At 12 weeks, 44% of patients on nivolumab achieved disease control compared to 50% in the nivolumab plus ipilimumab group.
The study successfully demonstrated anti-cancer activity without unexpected toxicity in relapsed patients with malignant pleural mesothelioma. Similar studies will likely lead to FDA approval for Opdivo and Yervoy as second-line treatments for mesothelioma.
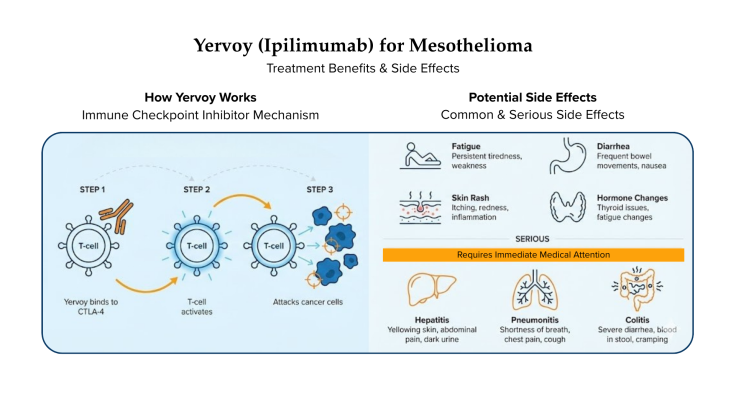
The potential benefits of Yervoy are not without the risk of side effects. Some side effects, such as fatigue, are minimal and go away when treatment ends. Other side effects are more serious, such as the risk of developing nerve problems or colitis (inflammation of the colon’s inner lining).
Patients on Yervoy and Opdivo may experience side effects of one drug or both, which may differ from the side effects of Yervoy alone.
Side effects of Yervoy may include:
People on combination therapy with Yervoy and Opdivo may have different reactions. Each immunotherapy drug has a list of potential side effects. Combination treatment can cause other unique adverse effects. Chemotherapy combined with Opdivo and Yervoy causes constipation in many people.
Bristol Myers Squibb cautions that the list of possible side effects may not be comprehensive.
Treating side effects early minimizes the risk of long-term injury. Tell your doctor immediately if you notice any changes to your body while taking Yervoy so they can offer quick and effective treatment. Patients can also report side effects directly to the FDA by calling 1-800-FDA-1088.
Yervoy is a checkpoint inhibitor immunotherapy drug that increases the effectiveness of the body’s T cells.
T cells are white blood cells. They are part of the immune system that fights infections and cellular abnormalities such as cancer. Yervoy inhibits the CTLA-4 protein that blocks immune response. It improves the ability of T cells to recognize and attack cancer cells. This mechanism increases your immune system’s natural defenses to help kill cancer cells.
Doctors administer Yervoy through an IV line in your vein or an access port. Pleural mesothelioma patients receive Yervoy every six weeks alongside Opdivo every three weeks for up to two years.
Immune checkpoint inhibitors now provide a viable alternative to chemotherapy for eligible patients. Further improvements will likely include new drugs and drug combinations to maximize benefits.
| YERVOY (IPILIMUMAB) INFORMATION | |
|---|---|
| Name | Yervoy |
| Alternate Names | Ipilimumab |
| Manufacturer | Bristol-Myers Squibb |
| Dosage | 1 mg/kg every six weeks |
| Administration Route | Intravenous |
| Active Ingredient | Ipilimumab |
| Drug Class | Monoclonal antibody, immune checkpoint inhibitor |
| Medical Code | J9228 |
| Interacting Drug | Warfarin, pradaxa, lovenox, heparin, eliquis, vemurafenib, cholera vaccine |
| Medical Studies | Ipilimumab and Nivolumab in the Treatment of Malignant Pleural Mesothelioma (INITIATE) |
| FDA Warning | Liver damage, thyroid dysfunction, fatigue, diarrhea, pruritus, nerve problems, eye problems, rash, colitis, fetal harm |

Discover new treatments with immunotherapy clinical trials near you. Get help with eligibility and enrollment.
Get StartedIt’s essential to discuss your medical history and any concerns with your doctor before taking Yervoy. For example, pregnancy is a severe contraindication, and Yervoy can harm your unborn baby. Patients on Yervoy should avoid breastfeeding during Yervoy treatment and for three months after the last dose.
Patients considering Yervoy should also provide a comprehensive list of their medications to their health care provider. Some drugs that bind to CTLA-4 can cause severe or fatal immune-mediated reactions. Potential drug interactions include certain vaccines (such as cholera), palifermin, Eliquis, Abraxane and Crestor.
Yervoy may cause complications in certain patients. Cytomegalovirus has been reported in patients with steroid-refractory immune-mediated colitis, a potential side effect of Yervoy. Ipilimumab should also be used cautiously in patients with a history of organ transplants, autoimmune disease or neurological disease.
Yes, the U.S. Food and Drug Administration approved Opdivo and Yervoy combination immunotherapy for first-line treatment of unresectable malignant pleural mesothelioma.
Opdivo is a checkpoint inhibitor that blocks PD-1, a pathway cancer cells use to hide from the immune system. Yervoy is a monoclonal antibody that targets cytotoxic T lymphocyte-associated antigen (CTLA-4). It is a protein that restricts the activity of the immune system. They prohibit cancer cell protection and elevate T-cell activity.
A 2021 clinical trial of Opdivo and Yervoy showed a median overall survival of 18.1 months. This compared with 14.1 months for patients on chemotherapy. The two-year survival rate for patients on Opdivo and Yervoy was 41%, compared to 27% for the chemotherapy group.
Your web browser is no longer supported by Microsoft. Update your browser for more security, speed and compatibility.
If you are looking for mesothelioma support, please contact our Patient Advocates at (855) 404-4592
The Mesothelioma Center at Asbestos.com has provided patients and their loved ones the most updated and reliable information on mesothelioma and asbestos exposure since 2006.
Our team of Patient Advocates includes a medical doctor, a registered nurse, health services administrators, veterans, VA-accredited Claims Agents, an oncology patient navigator and hospice care expert. Their combined expertise means we help any mesothelioma patient or loved one through every step of their cancer journey.
More than 30 contributors, including mesothelioma doctors, survivors, health care professionals and other experts, have peer-reviewed our website and written unique research-driven articles to ensure you get the highest-quality medical and health information.
My family has only the highest compliment for the assistance and support that we received from The Mesothelioma Center. This is a staff of compassionate and knowledgeable individuals who respect what your family is experiencing and who go the extra mile to make an unfortunate diagnosis less stressful. Information and assistance were provided by The Mesothelioma Center at no cost to our family.LashawnMesothelioma patient’s daughter


Marchese, S. (2026, March 9). Yervoy (Ipilimumab). Asbestos.com. Retrieved June 1, 2026, from https://www.asbestos.com/treatment/immunotherapy/yervoy/
Marchese, Sean. "Yervoy (Ipilimumab)." Asbestos.com, 9 Mar 2026, https://www.asbestos.com/treatment/immunotherapy/yervoy/.
Marchese, Sean. "Yervoy (Ipilimumab)." Asbestos.com. Last modified March 9, 2026. https://www.asbestos.com/treatment/immunotherapy/yervoy/.

Dr. Landau is the Medical Director of Virtual Hematology at the Medical University of South Carolina, where he leads programs that expand access to cancer care through telehealth. With more than 18 years of experience in oncology and hematology, he specializes in hematologic and genitourinary cancers, including bladder, prostate and kidney cancers. He has held multiple leadership roles in cancer program development and previously served as section chief of hematology and oncology at Orlando Health UF Health Cancer Center, where he founded its telehealth program.
Our fact-checking process begins with a thorough review of all sources to ensure they are high quality. Then we cross-check the facts with original medical or scientific reports published by those sources, or we validate the facts with reputable news organizations, medical and scientific experts and other health experts. Each page includes all sources for full transparency.
Please read our editorial guidelines to learn more about our content creation and review process.
