
Discover new treatments with immunotherapy clinical trials near you. Get help with eligibility and enrollment.
Get StartedImmune checkpoint inhibitors are a type of immunotherapy for mesothelioma. Cancer uses the body’s “off switches” to hide. These drugs block those switches, helping the immune system find and attack the cancer. They give people more options and hope.
Immune checkpoint inhibitors are a type of immunotherapy that treats pleural mesothelioma. They help your immune system find and fight this aggressive cancer. Mesothelioma cells use a special signal (PD-L1) to connect to a protein (PD-1). This protein works like an off switch on T cells, a key part of your body’s defense system.
Blocking these signals, immune checkpoint inhibitors help T cells stay active. Immunotherapies like Opdivo (nivolumab) and Keytruda (pembrolizumab) block PD-1. Similarly, Yervoy (ipilimumab) and Imjudo (tremelimumab) block CTLA-4. This protein turns off T cells earlier in the body’s immune response.
Experts classify checkpoint inhibitors into groups based on their targets. For example, PD-1 inhibitors stop the PD-1 signal. This helps T cells find and attack mesothelioma cells.
Checkpoint Inhibitors for Mesothelioma
The most common drugs are Keytruda, Opdivo and Yervoy, which are all Food and Drug Administration-approved for first-line or second-line unresectable pleural mesothelioma. These drugs are also monoclonal antibodies. They’re man-made and work like the body’s natural antibodies, which help fight disease.
Other checkpoint inhibitors are being studied. These include: Bavencio (avelumab), Imfinzi (durvalumab), Imjudo (tremelimumab) and Tecentriq (atezolizumab). All are PD-1 inhibitors, except for Imjudo, which is a CTLA-4 inhibitor.
One of the most common signals mesothelioma takes over is the PD-1/PD-L1 pathway. Checkpoint inhibitors block mesothelioma tumors from connecting with these proteins to “turn off” the immune system. These drugs turn T cells back on.
When T cells are turned back on, they can kill mesothelioma cells. This type of treatment helps the immune system fight the cancer. It’s a good option when the disease is advanced and surgery can’t be done.
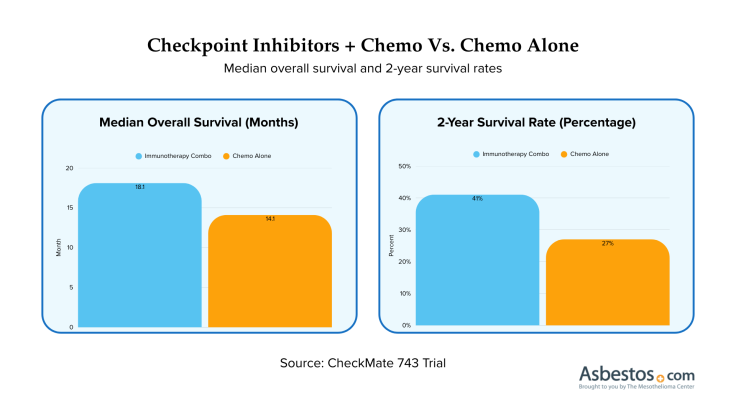
Checkpoint inhibitors can help people with pleural mesothelioma that can’t be removed with surgery. They may help people live longer and shrink the tumor. The results are different for each person and can depend on things like genes and overall health. Success seen in key clinical trials led to FDA approvals.
Efficacy of Checkpoint Inhibitors for Mesothelioma
Checkpoint inhibitors can also help people with sarcomatoid mesothelioma. This is a mesothelioma subtype that usually doesn’t respond to chemo. In some cases, these drugs can even double survival. The type of mesothelioma you have helps doctors choose the best treatment for you.
Opdivo and Yervoy are FDA-approved as a first-line treatment for pleural mesothelioma that can’t be surgically removed. For some patients, however, particularly those with advanced cancer or the sarcomatoid subtype of mesothelioma, treatment with checkpoint inhibitors focuses on palliative care, extending life and managing symptoms rather than curing the cancer.

Discover new treatments with immunotherapy clinical trials near you. Get help with eligibility and enrollment.
Get StartedPeople need to be in good health to get checkpoint inhibitor treatment. It’s considered risky for people with autoimmune diseases. The cell type of mesothelioma you have also matters. People with the epithelioid type often respond better.
“Because the FDA has approved this as a first-line mesothelioma treatment, this is something patients can be asking their doctor about,” said Dr. Catherine Perrault, a board-certified family physician and medical officer at The Mesothelioma Center. “It’s really, really important for us to take into account the different mesothelioma cell types.”
Eligibility Criteria for Checkpoint Inhibitors
Checkpoint inhibitors are FDA-approved as a first-line therapy or after chemo. They’re sometimes used off-label for peritoneal mesothelioma or are available through clinical trials.
Checkpoint inhibitors for mesothelioma turn on the immune system to fight cancer. Because of this, they can cause immune-related side effects like rash and swelling. Medications are available to manage most side effects.
Medicines like corticosteroids can help with some side effects. Doctors can also change the treatment if needed. Patients should tell their doctors about any side effects right away.
Common Side Effects of Checkpoint Inhibitors
Patients often wonder how checkpoint inhibitor side effects compare to chemo side effects. Chemo drugs like cisplatin and pemetrexed can cause nausea, hair loss and bone marrow suppression, which affect the whole body. Checkpoint inhibitors produce immune-related side effects such as fatigue and rash.
Many people with mesothelioma report feeling better overall on immunotherapy than on chemo, though this varies for each person. With reports of better tolerability of checkpoint inhibitors than with chemo, many patients are interested in it. Speak with your doctor about your eligibility.
Serious side effects are rare. But they can happen when mesothelioma and inflammation cause problems. For example, inflammation in the lungs can make it harder to breathe.
Rare But Serious Side Effects
These side effects require early intervention. It’s important to report any new or worsening symptoms to your doctor right away. With treatment, many of these serious side effects are reversible. However, they can leave permanent damage if left untreated.

Connect with top-rated mesothelioma specialists at a cancer center near you, who will personalize treatment options based on your diagnosis.
Find Your CenterMesothelioma clinical trials continue to explore checkpoint inhibitors in new ways. Clinical trials let patients try new drug combinations to help fight mesothelioma better. Many of these trials are currently recruiting participants.
Active Clinical Trials for Mesothelioma
Check with your mesothelioma doctor to see if clinical trials are enrolling near you. They can help determine if you’re eligible and could benefit you.
Checkpoint inhibitors aren’t a last resort. The FDA has approved Opdivo (nivolumab) combined with Yervoy (ipilimumab) as a first-line treatment for pleural mesothelioma that can’t be surgically removed. Doctors consider a person’s overall health, performance status and autoimmune history when determining eligibility. For people who aren’t candidates upfront, immunotherapy may still be an option after chemo.
Yes. Doctors can pause or stop checkpoint inhibitor treatment if serious side effects happen. Many side effects get better with quick care, and some people can start treatment again if it’s safe.
There isn’t a set end date for checkpoint inhibitor therapy. In the CheckMate 743 trial, patients received Opdivo every 3 weeks and Yervoy every 6 weeks, with treatment continuing until the cancer progressed, side effects became unmanageable or a complete response occurred. Some patients stay on treatment for 2 years or longer. Your oncologist will reassess your response at each cycle and adjust accordingly.
Doctors check your progress during treatment using scans like CT or PET and blood tests. If the tumor gets smaller, stays the same or grows more slowly, the treatment may be working. Your doctor will also watch your symptoms and overall health.
Patients also often notice physical signs that treatment may be working, such as improved breathing, less chest tightness, increased energy and reduced pain or fluid buildup. Some patients do experience pseudoprogression, where scans temporarily show what looks like tumor growth before a real response occurs. Always discuss scan results with your oncologist before drawing conclusions.
Yes. Some tests check for things like PD-L1 levels or other signs in the tumor to see if treatment might work. These tests can help, but they’re not always right. Even people with low PD-L1 levels can still get good results from treatment.
Checkpoint inhibitors can sometimes make immune system problems worse. They can cause swelling in places like the lungs. People with these issues should talk with their cancer doctor. In many cases, treatment can be changed to help prevent serious side effects.
After finishing checkpoint inhibitor therapy, patients typically enter a monitoring phase with follow-up scans every 3 months during the first year. Immune-related side effects can persist for weeks or months after the last infusion, so patients should keep reporting any new symptoms to their care team. Signs of recurrence to watch for include new or returning shortness of breath, chest pain, fatigue and fluid buildup. If the cancer returns, your oncologist may recommend maintenance therapy or a different treatment approach.
Your web browser is no longer supported by Microsoft. Update your browser for more security, speed and compatibility.
If you are looking for mesothelioma support, please contact our Patient Advocates at (855) 404-4592
The Mesothelioma Center at Asbestos.com has provided patients and their loved ones the most updated and reliable information on mesothelioma and asbestos exposure since 2006.
Our team of Patient Advocates includes a medical doctor, a registered nurse, health services administrators, veterans, VA-accredited Claims Agents, an oncology patient navigator and hospice care expert. Their combined expertise means we help any mesothelioma patient or loved one through every step of their cancer journey.
More than 30 contributors, including mesothelioma doctors, survivors, health care professionals and other experts, have peer-reviewed our website and written unique research-driven articles to ensure you get the highest-quality medical and health information.
My family has only the highest compliment for the assistance and support that we received from The Mesothelioma Center. This is a staff of compassionate and knowledgeable individuals who respect what your family is experiencing and who go the extra mile to make an unfortunate diagnosis less stressful. Information and assistance were provided by The Mesothelioma Center at no cost to our family.LashawnMesothelioma patient’s daughter


Asbestos.com. (2026, April 20). Immune Checkpoint Inhibitors for Mesothelioma. Retrieved June 4, 2026, from https://www.asbestos.com/treatment/immunotherapy/checkpoint-inhibitors/
"Immune Checkpoint Inhibitors for Mesothelioma." Asbestos.com, 20 Apr 2026, https://www.asbestos.com/treatment/immunotherapy/checkpoint-inhibitors/.
Asbestos.com. "Immune Checkpoint Inhibitors for Mesothelioma." Last modified April 20, 2026. https://www.asbestos.com/treatment/immunotherapy/checkpoint-inhibitors/.

Dr. Landau is the Medical Director of Virtual Hematology at the Medical University of South Carolina, where he leads programs that expand access to cancer care through telehealth. With more than 18 years of experience in oncology and hematology, he specializes in hematologic and genitourinary cancers, including bladder, prostate and kidney cancers. He has held multiple leadership roles in cancer program development and previously served as section chief of hematology and oncology at Orlando Health UF Health Cancer Center, where he founded its telehealth program.
Our fact-checking process begins with a thorough review of all sources to ensure they are high quality. Then we cross-check the facts with original medical or scientific reports published by those sources, or we validate the facts with reputable news organizations, medical and scientific experts and other health experts. Each page includes all sources for full transparency.
Please read our editorial guidelines to learn more about our content creation and review process.
