
Connect with top-rated mesothelioma specialists at a cancer center near you, who will personalize treatment options based on your diagnosis.
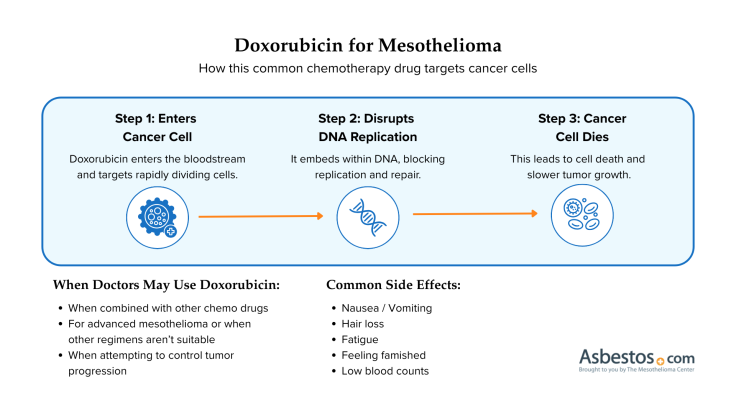
Find Your CenterFor more than 50 years, oncologists prescribed doxorubicin for the treatment of numerous cancers, including mesothelioma. Although the drug is highly effective, its use is limited by its potential to damage heart muscle tissue. It is commonly used in combination with other chemotherapy drugs.
Doxorubicin was approved by the U.S. Food and Drug Administration (FDA) in 1995 and is used to treat a number of cancers, including breast cancer and lung cancer.
Study results show that the drug can extend the lives of mesothelioma patients. The results show even more improved survival times when it is used along with other chemotherapy drugs, but using such a combination has potential to cause long-term side effects.
It is important to consider the benefits and risks of chemotherapy with the guidance of your oncologist and the support of your loved ones.
Doxorubicin is administered intravenously. The duration of administration and the dosage will depend upon whether it is combined with another chemotherapy drug.
Some studies are also testing it in a heated chemotherapy capacity. In heated chemotherapy, surgeons put the heated drug inside the chest or abdominal cavity immediately following surgery to enhance contact with cancer growth.
A 2021 clinical research study concluded that the combination of cisplatin and doxorubicin is the recommended drug regimen for hyperthermic intraperitoneal chemotherapy.
Several forms of doxorubicin, including Adriamycin and Doxil, have been tested on people with mesothelioma. Doxorubicin hydrochloride (HCL) is sold under the brand name Adriamycin and is manufactured by Pfizer. Doxorubicin HCL liposome injection is sold under the brand name Doxil in the U.S. and as Caelyx in the U.K. and is manufactured by Janssen.
| Name | Doxorubicin |
| Alternate Names | Adriamycin, Doxil, Caelyx, Doxorubicin Hydrochloride, Doxorubicin HCL, Rubex, Myocet |
| Manufacturer | Pfizer, Janssen Medical, Teva Parenteral Medicines, Bedford Laboratories, APP Pharmaceuticals |
| Dosage | 75 mg/m² every three weeks |
| Administration Route | Infusion |
| Active Ingredient | Doxorubicin HCL |
| Drug Class | Anthracycline |
| Medical Code | J9000, J9002, Q2049, Q2050 |
| Related Drug | Daunorubicin |
| Interacting Drug | Progesterone, turmeric, St. John’s wort, paclitaxel, anti-seizure medications, calcium channel blockers, azole antifungals, rifamycins, verapamil, cyclosporine, dexrazoxane, cytarabine, sorafenib, cyclophosphamide, digoxin, streptozocin, saquinavir, stavudine, trastuzumab, zidovudine |
| Medical Studies | Clinical Trial of Intraperitoneal Hyperthermic Chemotherapy (HIPEC/IPHC) |
| FDA Warning | Heart toxicity, nausea and vomiting, secondary cancer, immune suppression, myelosuppression, phlebitis/thrombophlebitis, liver damage, fetal harm |

Connect with top-rated mesothelioma specialists at a cancer center near you, who will personalize treatment options based on your diagnosis.
Find Your CenterThe most common side effects of doxorubicin include hair loss, darkening of nails, nausea, vomiting, bruising, abnormal heartbeat and stomach pain. If you feel pain, itching, redness, swelling, blisters or sores near the injection site following administration, inform your doctor promptly.
Patients treated with doxorubicin also have a slightly elevated risk of developing leukemia or myelodysplastic syndrome (which can develop into leukemia) years after treatment. The risk of leukemia increases in patients treated with a combination of doxorubicin and other chemotherapy drugs. In a review of 8,563 breast cancer patients who received treatment with doxorubicin and cyclophosphamide, 45 developed leukemia or myelodysplastic syndrome.
According to a review of mesothelioma clinical trials published in the journal Cancer, doxorubicin administered alone resulted in a median life span of seven to nine months. By combining it with other chemotherapy agents, median life span increases to seven to 13 months. Some people with mesothelioma have surpassed the median life expectancy by several years after combination chemotherapy. Patients with the epithelial cell type of mesothelioma tend to respond better to doxorubicin than patients with other cell types.
In one study, 67 peritoneal mesothelioma patients were treated with doxorubicin and the chemotherapy drugs paclitaxel and cisplatin. The patients achieved a median survival of 79 months, or about 6.5 years, and one patient lived past 12 years.
A small Japanese study on peritoneal mesothelioma patients published in 2010 confirmed these results. It studied the efficacy of the chemotherapy drug cisplatin alone and in combination with other chemotherapy agents. The study found that cisplatin was most effective in mesothelioma patients when it was combined with doxorubicin rather than with gemcitabine or pemetrexed, two other successful chemotherapy drugs.
An interesting case report of a nine-year survivor with pleural mesothelioma who responded well to combination chemotherapy with doxorubicin was published in 2012. The 67-year-old man was treated with the chemotherapy drugs etoposide, paclitaxel and pegylated liposomal doxorubicin hydrochloride (a form of doxorubicin that is more absorbed by tumors than normal tissues). The patient’s follow-up continues, and CT scans indicate the cancer hasn’t progressed for nine years.
Research continues to investigate the therapeutic benefits of doxorubicin for people with mesothelioma. Clinical trials in the United States and in other countries are ongoing.
Recommended ReadingYour web browser is no longer supported by Microsoft. Update your browser for more security, speed and compatibility.
If you are looking for mesothelioma support, please contact our Patient Advocates at (855) 404-4592
The Mesothelioma Center at Asbestos.com has provided patients and their loved ones the most updated and reliable information on mesothelioma and asbestos exposure since 2006.
Our team of Patient Advocates includes a medical doctor, a registered nurse, health services administrators, veterans, VA-accredited Claims Agents, an oncology patient navigator and hospice care expert. Their combined expertise means we help any mesothelioma patient or loved one through every step of their cancer journey.
More than 30 contributors, including mesothelioma doctors, survivors, health care professionals and other experts, have peer-reviewed our website and written unique research-driven articles to ensure you get the highest-quality medical and health information.
My family has only the highest compliment for the assistance and support that we received from The Mesothelioma Center. This is a staff of compassionate and knowledgeable individuals who respect what your family is experiencing and who go the extra mile to make an unfortunate diagnosis less stressful. Information and assistance were provided by The Mesothelioma Center at no cost to our family.LashawnMesothelioma patient’s daughter


Selby, K. (2026, March 9). Doxorubicin. Asbestos.com. Retrieved May 22, 2026, from https://www.asbestos.com/treatment/drugs/doxorubicin/
Selby, Karen. "Doxorubicin." Asbestos.com, 9 Mar 2026, https://www.asbestos.com/treatment/drugs/doxorubicin/.
Selby, Karen. "Doxorubicin." Asbestos.com. Last modified March 9, 2026. https://www.asbestos.com/treatment/drugs/doxorubicin/.

Dr. Rupesh Kotecha is a renowned radiation oncologist in leadership roles at Miami Cancer Institute. He is an associate professor at Florida International University's college of medicine and an adjunct faculty member at Memorial Sloan Kettering.
Our fact-checking process begins with a thorough review of all sources to ensure they are high quality. Then we cross-check the facts with original medical or scientific reports published by those sources, or we validate the facts with reputable news organizations, medical and scientific experts and other health experts. Each page includes all sources for full transparency.
Please read our editorial guidelines to learn more about our content creation and review process.
