Mesothelioma
Mesothelioma is a cancer that forms tumors in the mesothelium, the thin tissue lining the lungs, heart, belly and testes. The most common type, pleural mesothelioma, affects the lung lining. Asbestos exposure causes mesothelioma. Surgery, immunotherapy, chemo and radiation are common treatments.
What Is Mesothelioma?
Mesothelioma is a rare cancer that develops in the thin layer of tissue called the mesothelium that lines your body’s cavities and many internal organs. The primary cause of mesothelioma is asbestos exposure.
Key Facts About Malignant Mesothelioma
- Symptoms can take 20 to 60 years to appear after asbestos exposure.
- Doctors diagnose fewer than 3,000 new cases of mesothelioma each year.
- Most mesothelioma patients are 75 to 79 years old at the time of diagnosis.
- Mesothelioma is usually cancerous (malignant). Noncancerous or benign cases have been reported.
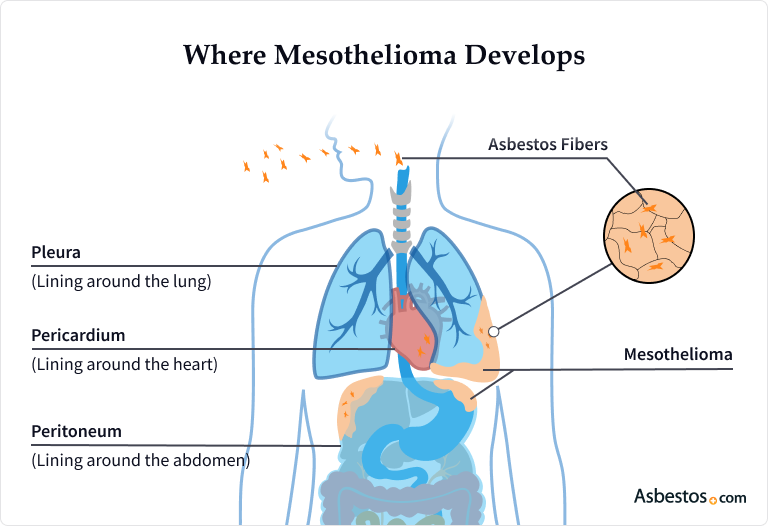
Doctors classify malignant mesothelioma into different types based on where tumors form (around the lungs, abdomen, heart or testes) in the mesothelium. Treatment options, outlook (prognosis) and life expectancy can vary among these different mesothelioma types.
Types of Mesothelioma Cancer
Mesothelioma cancer forms in the lining of the lungs, abdomen, heart or testes. The most common type of mesothelioma is called pleural mesothelioma because it forms in the pleura.
Of all mesothelioma cases, 70% to 79% are pleural mesothelioma. People with pleural mesothelioma can develop tumors on the lung lining, chest wall, diaphragm (the muscle that helps you breathe) and other parts of the chest.
The second most common type is called peritoneal mesothelioma because it forms in the peritoneum. People with peritoneal mesothelioma can develop tumors on the lining of abdominal organs and the gut wall. The rarest types are testicular mesothelioma and pericardial mesothelioma.
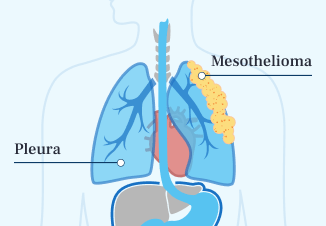
- Pleural Mesothelioma
-
The most common type of mesothelioma is pleural mesothelioma, also known as malignant pleural mesothelioma. It forms in the pleura, the protective lining around the outside of the lungs.
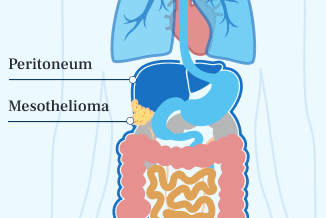
- Peritoneal Mesothelioma
-
Peritoneal mesothelioma, the second most common type, forms in the abdominal lining. It has a much higher survival rate. Surgery often works better for this type.
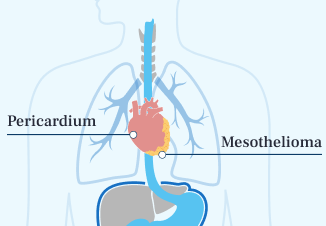
- Pericardial Mesothelioma
-
Pericardial mesothelioma affects about 1% of patients. Treatment options are much more limited. The median survival is 2 to 6 months.
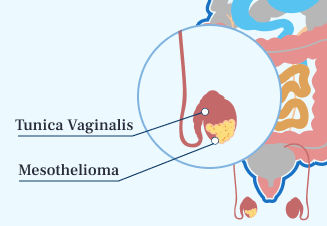
- Testicular Mesothelioma
-
The rarest type is testicular mesothelioma. Treatment options are much more limited. About one-third of patients live for at least 10 years.
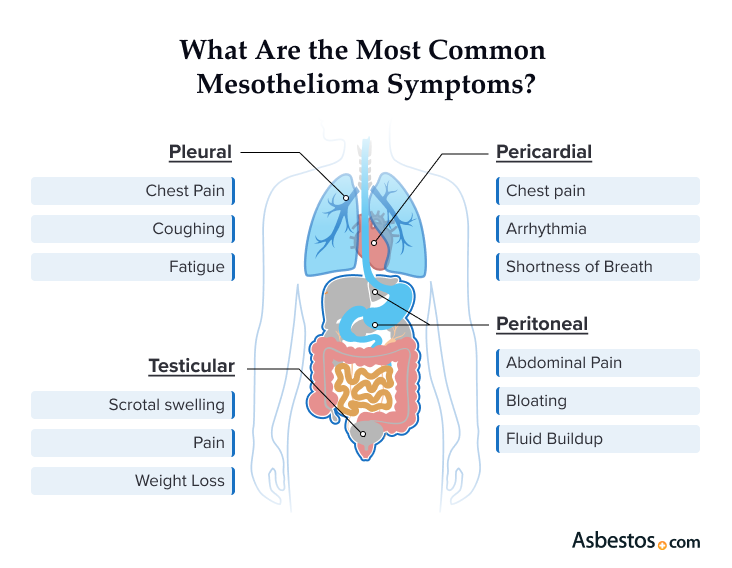
Symptoms of Mesothelioma
The most common symptoms of mesothelioma include fatigue, weight loss and fever. Symptoms of mesothelioma vary based on where the cancer forms in your body.
It usually takes 20 to 60 years after your first exposure to asbestos for symptoms to appear. Experts call this the mesothelioma latency period.
Symptoms by Mesothelioma Type
- Pleural mesothelioma: chest pain, fatigue, fluid buildup (pleural effusion), persistent cough, shortness of breath and unexplained weight loss
- Peritoneal mesothelioma: abdominal pain or swelling, fluid buildup (ascites), constipation or diarrhea, loss of appetite, weight loss and nausea or vomiting
- Pericardial mesothelioma: chest pain, difficulty breathing, fatigue and irregular heartbeat
- Testicular mesothelioma: pain or discomfort in the groin area and swelling or lumps in the testes
These symptoms can be unclear and look like other, less serious issues. If you have mesothelioma symptoms, see a specialist for help. Early diagnosis can improve your outlook and help you live longer.

Understand your diagnosis, top doctors and ways to afford care.
Get Your Free GuideWhat Causes Mesothelioma?
Asbestos exposure causes mesothelioma. People can breathe in asbestos fibers from damaged products. Dr. Marcelo DaSilva, chief of thoracic surgery at AdventHealth Cancer Institute, says these inhaled fibers then cause inflammation.
“It causes chronic inflammation in the mesothelium,” Dr. DaSilva tells us. “This can lead to genetic changes.” These changes to the DNA in your cells can cause them to become mesothelioma cancer cells. These mutations are often when tumors begin to grow.
Who Is Most at Risk of Mesothelioma?
People who are regularly exposed to asbestos in the workplace are most at risk for mesothelioma. But no amount of asbestos exposure is considered safe.
There are also other factors that may increase the risk that someone exposed to asbestos may develop mesothelioma. These include family history, gender, age and overall health.
Mesothelioma Risk Factors
- Environmental exposure: People living near asbestos mines or factories might inhale asbestos particles in the air or ground.
- Family history of mesothelioma: Family members who inherit the BAP1 mutation are sensitive to asbestos exposure. They have a higher risk of mesothelioma with exposure.
- Occupational exposure: Military personnel and workers in industries such as construction, shipbuilding, automotive manufacturing and mining were often exposed to asbestos fibers.
- Secondary exposure: Family members of workers exposed to asbestos can also be at risk if asbestos fibers were brought home on clothing, tools, hair and skin.
If any of these risk factors apply to you or a loved one, talk to your doctor about mesothelioma screenings. X-rays and other tests can help catch mesothelioma early. Tell your doctor about your exposure and ask how frequently you should be screened.
I would go down there with [my father], and it was very dusty in the boiler room. There would be chunks of gray, fibrous stuff down there. I didn’t know what it was, but I’d pick it up and play with it.
How Is Mesothelioma Diagnosed?
Doctors diagnose mesothelioma using a combination of tests, such as a physical exam and biopsy. It’s a hard cancer to diagnose because the symptoms can be a lot like other, more common illnesses.
A diagnosis confirms details such as your cancer type and stage or how far your cancer has progressed. These details help your doctor create the best treatment plan for you.
Methods to Diagnose Mesothelioma
- Physical examination: Doctors usually start by checking for lumps or other unusual signs. They then use lung function tests to check lung health.
- Imaging tests: X-rays, CT scans, MRIs and PET scans help doctors see possible abnormalities in the body.
- Blood tests: Doctors check for specific proteins or biomarkers in the blood. These can help indicate cancer may be mesothelioma.
- Biopsy: This is the definitive way doctors diagnose mesothelioma. Pathologists take a small sample of tissue and examine it under a microscope for cancer cells.
Pathologists also check to see what mesothelioma cell types are present. They may be epithelioid, sarcomatoid and biphasic. Each cell type can respond differently to specific therapies. Knowing your cell type helps your doctor pick the best treatment plan for you. It also helps them assess your outlook or prognosis.
In 2025, the American Society of Clinical Oncology recommended genetic testing for mesothelioma. It can show mutations that impact treatment. For example, patients with a BAP1 mutation live longer with chemotherapy.
| % Patients Diagnosed | % of Male | % of Female | |
|---|---|---|---|
| Stage I | 18% | 18% | 24% |
| Stage II | 11% | 11% | 12% |
| Stage III | 23% | 23% | 20% |
| Stage IV | 48% | 48% | 44% |
Staging Mesothelioma
The stages of mesothelioma describe how far your cancer has progressed, specifically tumor size, distance from the point of origin and location. As tumors grow and spread to distant organs, the stages advance. Stages 1 and 2 are early stages. And stages 3 and 4 are late stages.
Only pleural mesothelioma has a formal staging system. Doctors use the TNM system, which stands for Tumor, Node and Metastasis. The stage of your cancer can affect the treatment options your doctor will recommend for you.

Connect with trusted specialists who truly care about your health. Get fast, stress-free appointment help.
Find a Doctor NowHow Is Mesothelioma Treated?
Doctors treat mesothelioma using surgery, chemo, radiation and immunotherapy. The American Society of Clinical Oncology reviews treatments. They create treatment guidelines and update them every year. According to our exclusive 2025 survey, chemo is the most common treatment, with 32% of respondents receiving it.
Your treatment options may vary depending on your mesothelioma type, including whether it’s cancerous or noncancerous. Your overall health will also play a role in determining the best therapies for you.
Types of Mesothelioma Treatments
- Chemotherapy
- Clinical trials
- Immunotherapy
- Multimodal therapy
- Palliative care
- Radiation therapy
- Surgery
- Tumor Treating Fields
Many patients switch to another treatment if one doesn’t show results. Pleural mesothelioma survivor Wally Rogers tells us chemo wasn’t working for him. He swapped chemo for immunotherapy and saw a remarkable recovery.
Wally’s CT scans showed no remaining tumor cells after immunotherapy. His health has improved a lot. Now he says he can hike and ski again. Wally recommends looking into every option to improve your outlook or prognosis.
Mesothelioma Prognosis
The outlook or prognosis for mesothelioma is generally poor, but it improves with treatment. Mesothelioma statistics show 12% of pleural patients live 5 years. The 5-year survival rate for people with the peritoneal type is 65%.
While the life expectancy for mesothelioma is about 12 months, your stage at diagnosis, treatments and your overall health will affect your individual prognosis. Treatment and a healthy lifestyle may lead to partial remission. Vaccines can help prevent serious lung infections. Talk to your doctor about ways to boost your prognosis.
As part of my treatment, I had surgery with HIPEC. Studies show HIPEC drastically improves life expectancy in about 50% of the peritoneal mesothelioma patients who have the procedure. It definitely worked in my favor and was a success!
Mesothelioma Prevention and Risk Reduction
Try to limit asbestos exposure to lower the risk of mesothelioma. You and your family can take steps to stay safe from exposure. Hire a licensed expert to remove asbestos. Don’t remove asbestos yourself.
Employers need to follow safety rules that prevent exposure at work. Giving the right personal protective equipment is a key part of these rules. Examples are special masks, gloves and protective clothing. These items help stop tiny asbestos fibers from getting into your lungs.
Always wear this protective gear when you’re near asbestos. If you see someone at work ignoring safety rules, or if the protective gear isn’t adequate, report it to your union representative. They can help make sure everyone stays safe. Speaking up helps protect everyone’s health.
If you’ve been exposed to asbestos, tell your doctor. You may need tests to detect cancer early. If you smoke, consider quitting. This is important, especially if you’re around asbestos. Smoking doesn’t cause mesothelioma. However, it raises the risk of lung cancer in people exposed to asbestos.
Living With Mesothelioma
Living with a rare cancer, such as mesothelioma, can be tough for patients and their caregivers. It’s normal to worry and feel stressed about how to move ahead.
Your Recommended Next Steps
- Explore clinical trials. They might improve your prognosis.
- Adopt a healthier lifestyle. Simple exercises and a balanced diet can ease your side effects. They can boost your mood while you undergo cancer treatment.
- Join a support group. It can help reduce anxiety, distress, fatigue and pain.
- Financial help covers medical costs. Asbestos trust funds also provide compensation.
- Veterans can access the VA for care and benefits. The VA also offers caregiver benefits and more.
Focus on getting excellent treatment. You can empower your family with knowledge, resources and hope. Peritoneal survivor Tamron Little tells us more resources would have helped her when she was first diagnosed.
“I would have looked for different mesothelioma resources, aid or even support groups,” Tamron shares with us. “That would have helped me while I was battling the disease.”
Commonly Asked Questions About Mesothelioma
- Can mesothelioma go into remission?
-
Yes, some mesothelioma patients experience remission. Remission may be partial or complete.
- How do you know if you have mesothelioma?
-
If you have a history of asbestos exposure, share this with your doctor. Let your doctor know if you have any ongoing symptoms. These include chest pain, a cough or trouble breathing.
Mesothelioma has a long latency period. It can take 20 to 60 years to develop after exposure. So symptoms can appear decades later.
- How is mesothelioma different from lung cancer?
-
Mesothelioma isn’t a form of lung cancer. But both diseases are cancers. Mesothelioma grows on the mesothelium, which lines the cavities of the body. It most commonly develops on the part of the mesothelium called the pleura, which is the lining that covers the lungs and chest cavity. Lung cancer develops within the lung tissue.
- Is there a cure for mesothelioma?
-
Mesothelioma isn’t curable. Researchers are investigating new treatments through clinical trials. Some patients live for years beyond what is expected. A small number of people have survived for more than a decade after diagnosis. Immunotherapy and other new treatments bring hope for a cure.






